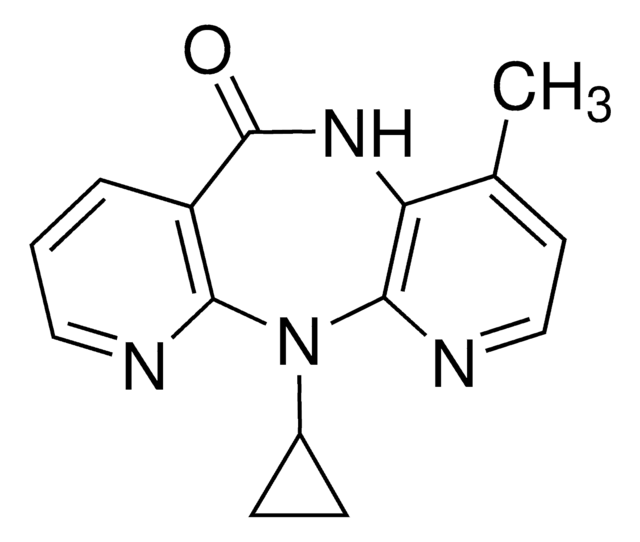
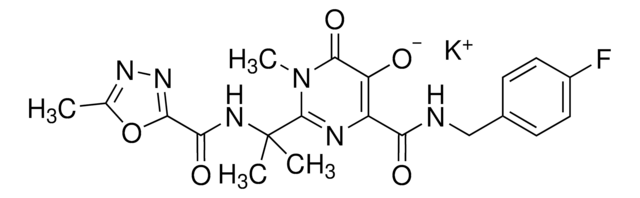
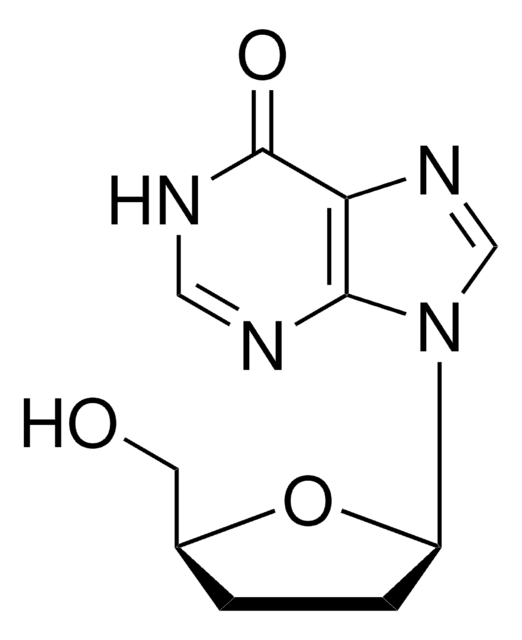
推荐产品
化驗
≥98% (HPLC)
形狀
powder
顏色
white to beige
溶解度
water: 10 mg/mL, clear
起源
GlaxoSmithKline
儲存溫度
room temp
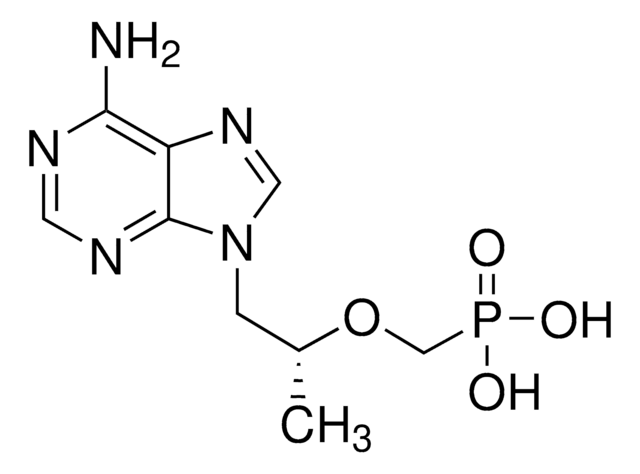
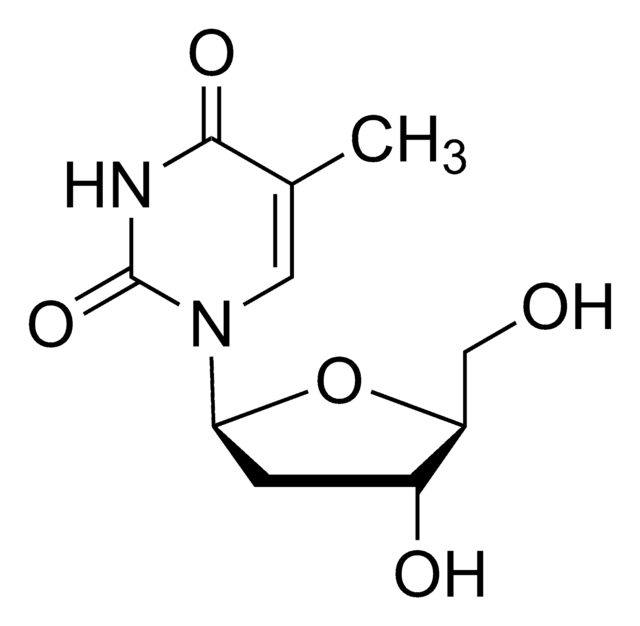
SMILES 字串
NC1=NC(=O)N(C=C1)[C@@H]2CS[C@H](CO)O2
InChI
1S/C8H11N3O3S/c9-5-1-2-11(8(13)10-5)6-4-15-7(3-12)14-6/h1-2,6-7,12H,3-4H2,(H2,9,10,13)/t6-,7+/m0/s1
InChI 密鑰
JTEGQNOMFQHVDC-NKWVEPMBSA-N
正在寻找类似产品? 访问 产品对比指南
應用
Lamivudine has been used to deplete the Hepatitis B Virus (HBV) covalently closed circular DNA (cccDNA) forms for the preparation of inverse nested PCR.
生化/生理作用
拉米夫定是一种有效的核苷类似物逆转录酶抑制剂(nRTI)。 它是胞苷的类似物,除抑制乙型肝炎的逆转录酶外,还可抑制两种类型(1型和2型)的HIV逆转录酶。它需要磷酸化为其三磷酸形式才具有活性。3TC-三磷酸也抑制细胞DNA聚合酶。
特點和優勢
This compound is a featured product for ADME Tox research. Click here to discover more featured ADME Tox products. Learn more about bioactive small molecules for other areas of research at sigma.com/discover-bsm.
This compound was developed by GlaxoSmithKline. To browse the list of other pharma-developed compounds and Approved Drugs/Drug Candidates, click here.
訊號詞
Warning
危險聲明
危險分類
Repr. 2
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
其他客户在看
Elaine Y Liu et al.
Cell reports, 27(5), 1409-1421 (2019-05-03)
Loss of the nuclear RNA binding protein TAR DNA binding protein-43 (TDP-43) into cytoplasmic aggregates is the strongest correlate to neurodegeneration in amyotrophic lateral sclerosis and frontotemporal degeneration. The molecular changes associated with the loss of nuclear TDP-43 in human
Chia-Yen Dai et al.
The Journal of antimicrobial chemotherapy, 68(10), 2332-2338 (2013-06-27)
For hepatitis B e antigen (HBeAg)-positive patients, continuing therapy (consolidation) for 6-12 months before cessation of nucleos(t)ide analogues (NAs) was recommended. This study aimed to investigate whether a longer period of lamivudine consolidation therapy leads to better outcomes and the
Raph L Hamers et al.
Journal of acquired immune deficiency syndromes (1999), 64(2), 174-182 (2013-07-31)
This study assessed HIV-hepatitis B virus (HBV) coinfection in southern Africa in terms of prevalence, viral characteristics, occult HBV, and the effect of lamivudine- versus tenofovir-containing first-line combination antiretroviral treatment (cART) on HBV-related outcomes. A multicenter prospective cohort of HIV-infected
Wai-Kay Seto et al.
Hepatology (Baltimore, Md.), 58(3), 923-931 (2013-03-08)
The profile and clinical significance of serum hepatitis B surface antigen (HBsAg) levels during long-term nucleoside analogue (NA) therapy in chronic hepatitis B (CHB) is undetermined. From 1994 to 2002, 322 Chinese CHB patients were started on lamivudine in our
Kiyoaki Ito et al.
Hepatology (Baltimore, Md.), 59(1), 89-97 (2013-07-31)
The proportion of patients who progress to chronicity following acute hepatitis B (AHB) varies widely worldwide. Moreover, the association between viral persistence after AHB and hepatitis B virus (HBV) genotypes in adults remains unclear. A nationwide multicenter study was conducted
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门