About This Item
meets purity specifications of JECFA
推荐产品
等級
FG
Fragrance grade
Halal
Kosher
natural
品質等級
agency
follows IFRA guidelines
meets purity specifications of JECFA
法律遵循
EU Regulation 1223/2009
EU Regulation 1334/2008 & 178/2002
FCC
FDA 21 CFR 117
蒸汽密度
3.04 (vs air)
蒸汽壓力
0.43 mmHg ( 20 °C)
化驗
≥99%
形狀
liquid
自燃溫度
824 °F
expl. lim.
10 %
折射率
n20/D 1.398 (lit.)
pH值
2 (25 °C, 10 g/L)
3 (20 °C, 10 g/L)
bp
162 °C (lit.)
mp
−6-−3 °C (lit.)
密度
0.964 g/mL at 25 °C (lit.)
正離子痕跡
As: ≤3 ppm
Cd: ≤1 ppm
Hg: ≤1 ppm
Pb: ≤10 ppm
應用
flavors and fragrances
文件
see Safety & Documentation for available documents
食物過敏原
no known allergens
香料過敏原
no known allergens
感官的
cheese; fruity; buttery
SMILES 字串
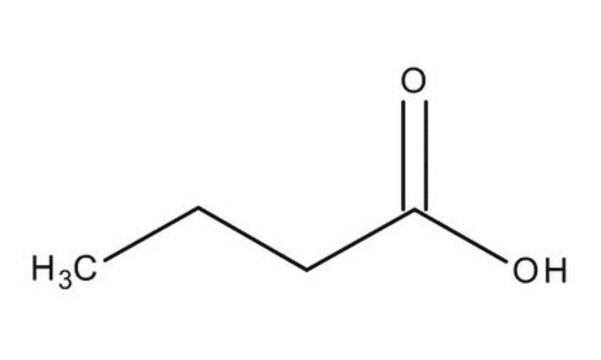
CCCC(O)=O
InChI
1S/C4H8O2/c1-2-3-4(5)6/h2-3H2,1H3,(H,5,6)
InChI 密鑰
FERIUCNNQQJTOY-UHFFFAOYSA-N
基因資訊
human ... HDAC1(3065)
正在寻找类似产品? 访问 产品对比指南
一般說明
訊號詞
Danger
危險聲明
危險分類
Acute Tox. 4 Oral - Eye Dam. 1 - Skin Corr. 1B - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
8A - Combustible corrosive hazardous materials
水污染物質分類(WGK)
WGK 1
閃點(°F)
161.6 °F - closed cup
閃點(°C)
72 °C - closed cup
個人防護裝備
Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter
其他客户在看
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系客户支持