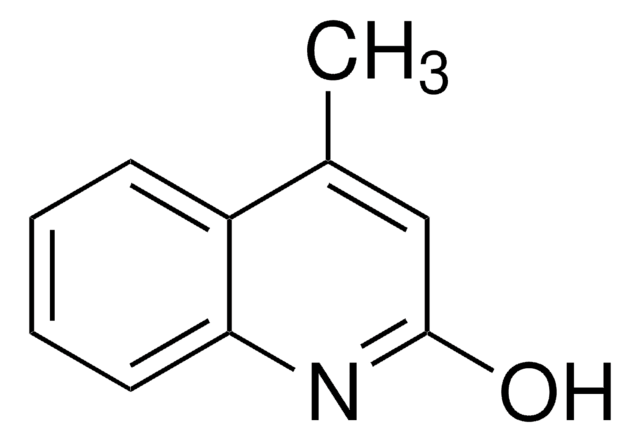
推荐产品
品質等級
化驗
98.5%
SMILES 字串
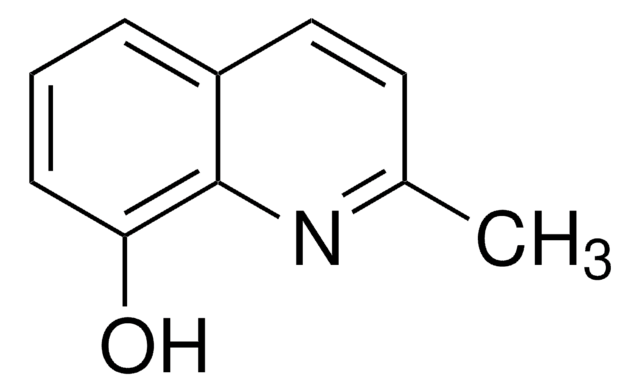
Cc1cc(O)c2ccccc2n1
InChI
1S/C10H9NO/c1-7-6-10(12)8-4-2-3-5-9(8)11-7/h2-6H,1H3,(H,11,12)
InChI 密鑰
NWINIEGDLHHNLH-UHFFFAOYSA-N
應用
4-Hydroxy-2-methylquinoline can be used as an intermediate in the synthesis of a wide range of medicinally important compounds such as:
- Synthesis of 2-(quinolin-4-yloxy)acetamides as potent antitubercular agents.
- Synthesis of 2-arylethenylquinoline derivatives for the treatment of Alzheimer′s disease.
- Synthesis of 1,10-diethoxy-1H-pyrano[4,3-b]quinolones as antibacterial agents.
- Synthesis of phenylimidazole-pyrazolo[1,5-c]quinazolines as potent phophodiesterase 10A (PDE10A) inhibitors.
訊號詞
Warning
危險聲明
危險分類
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
dust mask type N95 (US), Eyeshields, Gloves
Synthesis and SAR study of new phenylimidazole-pyrazolo [1, 5-c] quinazolines as potent phosphodiesterase 10A inhibitors.
Asproni B, et al.
Bioorganic & Medicinal Chemistry, 19(1), 642-649 (2011)
Synthesis of new 1, 10-diethoxy-1H-pyrano [4, 3-b] quinolines and their antibacterial studies.
Dhanabal T, et al.
Indian J. Chem. B, 45B(02) (2006)
2-(Quinolin-4-yloxy) acetamides are active against drug-susceptible and drug-resistant Mycobacterium tuberculosis strains.
Pissinate K, et al.
ACS Medicinal Chemistry Letters, 7(3), 235-239 (2016)
Quinaldine derivatives: Preparation and biological activity.
Jampilek J, et al.
Medicinal Chemistry, 1(6), 591-599 (2005)
Design, synthesis, and biological evaluation of 2-arylethenylquinoline derivatives as multifunctional agents for the treatment of Alzheimer's disease.
Wang X Q, et al.
European Journal of Medicinal Chemistry, 89, 349-361 (2015)
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系客户支持