推荐产品
化驗
98%
形狀
solid
mp
155-157 °C (lit.)
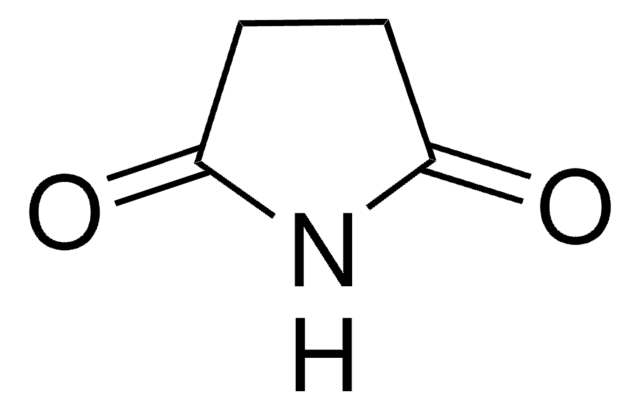
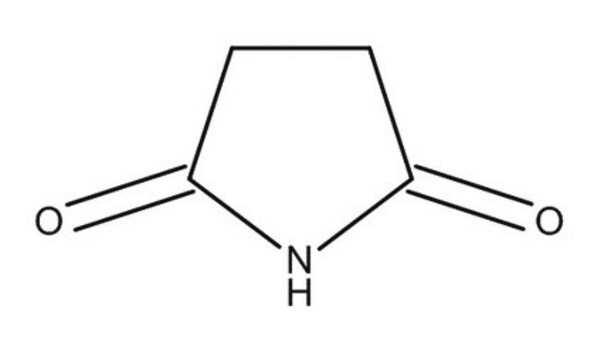
SMILES 字串
O=C1CCCC(=O)N1
InChI
1S/C5H7NO2/c7-4-2-1-3-5(8)6-4/h1-3H2,(H,6,7,8)
InChI 密鑰
KNCYXPMJDCCGSJ-UHFFFAOYSA-N
一般說明
A glutarimide antibiotic, 9-methylstreptimidone, shows antiviral, antitumor and antifungal activities.
應用
Reactant for:
Thionations
Biocatalytic asymmetric synthesis of chiral amines from ketones applied to sitagliptin manufacture
Synthesis of β-adrenoceptor ligands
Enantioselective synthesis of securinega alkaloids
Intramolecular amidocyclopropanation reactions
Synthesis of alpha-fluoro-alpha amino amides
Thionations
Biocatalytic asymmetric synthesis of chiral amines from ketones applied to sitagliptin manufacture
Synthesis of β-adrenoceptor ligands
Enantioselective synthesis of securinega alkaloids
Intramolecular amidocyclopropanation reactions
Synthesis of alpha-fluoro-alpha amino amides
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
其他客户在看
Alexander A Bisset et al.
Chemical communications (Cambridge, England), 48(98), 11978-11980 (2012-11-07)
The synthesis of (3E)-1-benzyl-3-[(2-oxopyridin-1(2H)-yl)methylidene]piperidine-2,6-dione 5 from N-benzylglutarimide was achieved in three steps. The asymmetric hydrogenation of 4 gave either the product of partial reduction (10) or full reduction (13), depending on the catalyst which was employed, in high ee in
Deevi Basavaiah et al.
Organic & biomolecular chemistry, 6(6), 1034-1039 (2008-03-11)
A simple and convenient synthesis of di(E)-arylidene-tetralone-spiro-glutarimides from Baylis-Hillman acetates via an interesting biscyclization strategy involving facile C-C and C-N bond formation is described. Also, one-pot multistep transformation of the Baylis-Hillman acetates into di(E)-arylidene-spiro-bisglutarimides is presented.
Pei-Qiang Huang et al.
Organic letters, 8(7), 1435-1438 (2006-03-28)
[reaction: see text] Using 5b as a common intermediate, the first asymmetric synthesis of (-)-epiquinamide (4) and a formal asymmetric synthesis of (-)-homopumiliotoxin 223G (2) is described. A key feature of our approach is the flexible introduction of a functionalized
Chuanjin Tian et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 18(45), 14305-14313 (2012-10-16)
The significance of the molecular chirality of drugs has been widely recognized due to the thalidomide tragedy. Most of the new drugs reaching the market today are single enantiomers, rather than racemic mixtures. However, many optically pure drugs, including thalidomide
Jianhua Ju et al.
Organic letters, 9(25), 5183-5186 (2007-11-14)
Lactimidomycin (LTM, 1) is a macrolide antitumor antibiotic with a glutarimide side chain from Streptomyces amphibiosporus ATCC53964. To further develop LTM and related analogues as drug candidates we have (i) improved LTM production by approximately 20 fold, (ii) identified three
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门