推荐产品
品質等級
化驗
99%
形狀
solid
bp
298-299 °C (lit.)
mp
68-70 °C (lit.)
溶解度
alcohol: freely soluble (hot)
diethyl ether: slightly soluble
water: insoluble
官能基
ester
phenoxy
phenyl
SMILES 字串
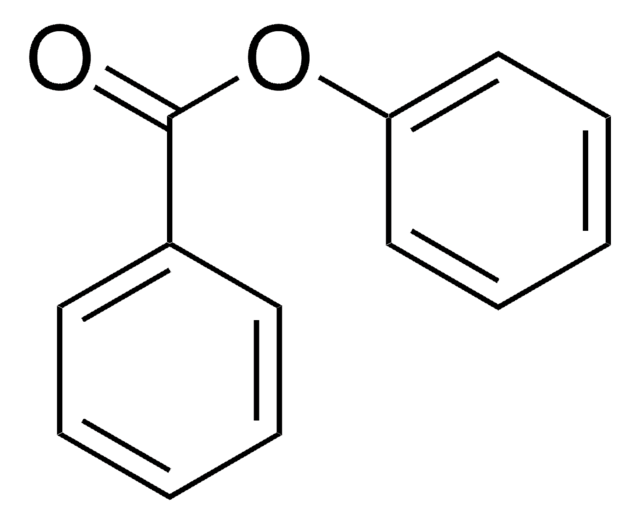
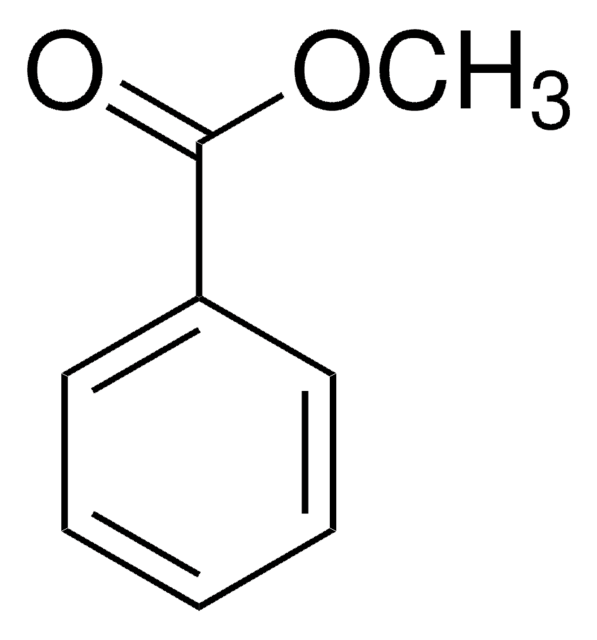
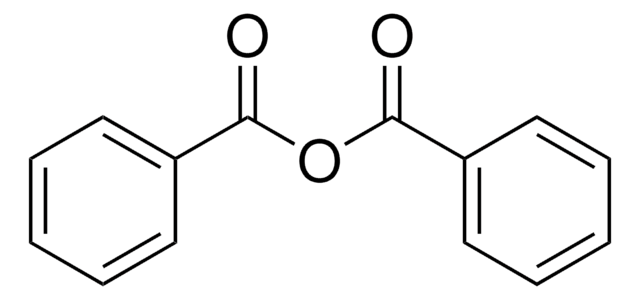
O=C(Oc1ccccc1)c2ccccc2
InChI
1S/C13H10O2/c14-13(11-7-3-1-4-8-11)15-12-9-5-2-6-10-12/h1-10H
InChI 密鑰
FCJSHPDYVMKCHI-UHFFFAOYSA-N
正在寻找类似产品? 访问 产品对比指南
一般說明
苯甲酸苯酯作为前体,通过分子内芳基-芳基偶联反应产生用于合成(−)五加酮的中间体。
應用
訊號詞
Warning
危險聲明
危險分類
Acute Tox. 4 Oral - Skin Irrit. 2
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
dust mask type N95 (US), Eyeshields, Gloves
其他客户在看
商品
The Fries rearrangement reaction is an organic name reaction which involves the conversion of phenolic esters into hydroxyaryl ketones on heating in the presence of a catalyst. Suitable catalysts for this reaction are Brønsted or Lewis acids such as HF, AlCl3, BF3, TiCl4, or SnCl4. The Fries rearrangement reaction is an ortho, para-selective reaction, and is used in the preparation of acyl phenols. This organic reaction has been named after German chemist Karl Theophil Fries.
The Fries rearrangement reaction is an organic name reaction which involves the conversion of phenolic esters into hydroxyaryl ketones on heating in the presence of a catalyst. Suitable catalysts for this reaction are Brønsted or Lewis acids such as HF, AlCl3, BF3, TiCl4, or SnCl4. The Fries rearrangement reaction is an ortho, para-selective reaction, and is used in the preparation of acyl phenols. This organic reaction has been named after German chemist Karl Theophil Fries.
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门