22620
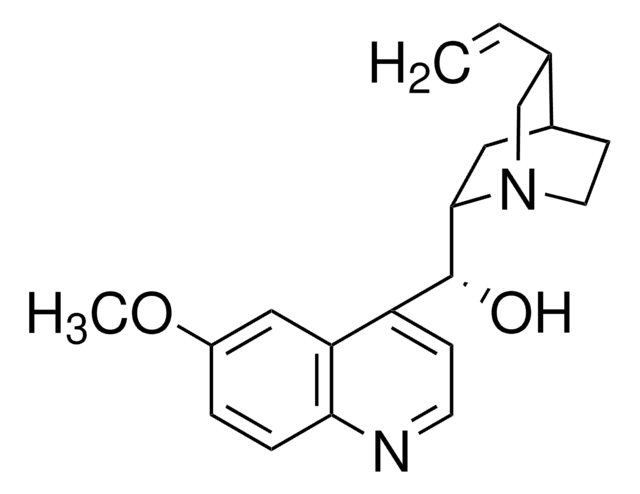
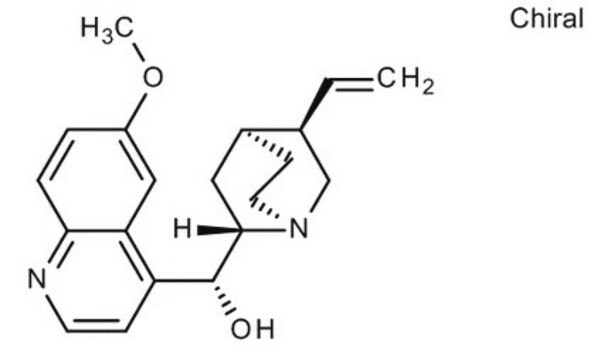
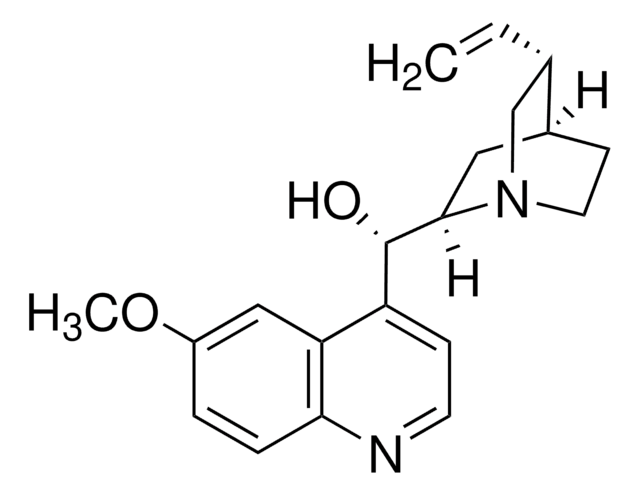
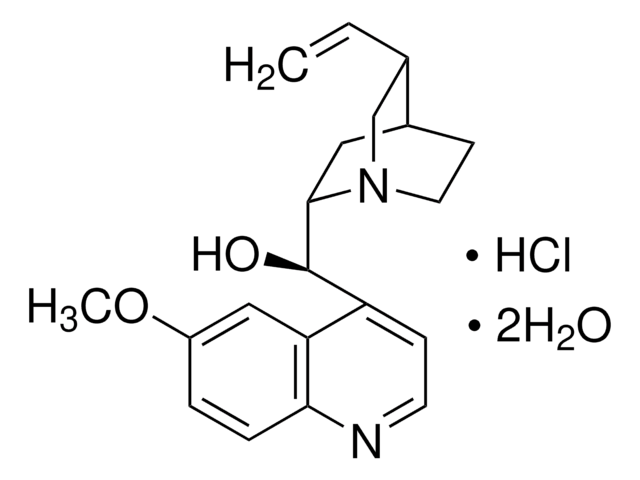
Quinine
suitable for fluorescence, anhydrous, ≥98.0% (dried material, NT)
Sinónimos:
6′-Methoxycinchonidine
About This Item
Productos recomendados
Nivel de calidad
Ensayo
≥98.0% (dried material, NT)
Formulario
powder
actividad óptica
[α]20/D −126±5°, c = 1% in chloroform
impurezas
≤5% dihydroquinine (HPLC)
pérdida
≤1% loss on drying, 110 °C
mp
173-175 °C (lit.)
solubilidad
H2O: soluble
fluorescencia
λex 347 nm; λem 448 nm in 0.5 M sulfuric acid
idoneidad
suitable for fluorescence
cadena SMILES
COc1ccc2nccc([C@@H](O)[C@@H]3C[C@@H]4CCN3C[C@@H]4C=C)c2c1
InChI
1S/C20H24N2O2/c1-3-13-12-22-9-7-14(13)10-19(22)20(23)16-6-8-21-18-5-4-15(24-2)11-17(16)18/h3-6,8,11,13-14,19-20,23H,1,7,9-10,12H2,2H3/t13-,14-,19-,20+/m0/s1
Clave InChI
LOUPRKONTZGTKE-WZBLMQSHSA-N
Información sobre el gen
human ... ABCB1(5243) , CYP2C9(1559) , CYP2D6(1565)
rat ... Cyp2d1(266684) , Cyp2d2(25053) , Cyp2d3(24303) , Cyp2d4v1(171522)
¿Está buscando productos similares? Visita Guía de comparación de productos
Descripción general
Aplicación
- To study its in vitro antimalarial activity in combination with omeprazole.
- To analyze its effect on viscosity and friction of saliva.
- As a test agent to study its impact on the accumulation of the fluorescent P-glycoprotein (Pgp) substrates in P-glycoprotein overexpressing breast cancer cells.
- To study its influence on the pyramidal cell intrinsic properties, extracellular potassium transients, and epileptiform activity in vitro.
- As a reference compound to identify alkaloids by phytochemical screening of Deianira erubescens, Strychnos pseudoquina and Remijia ferruginea plants.
Acciones bioquímicas o fisiológicas
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Acute Tox. 4 Oral - Skin Sens. 1
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 1
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico