227269
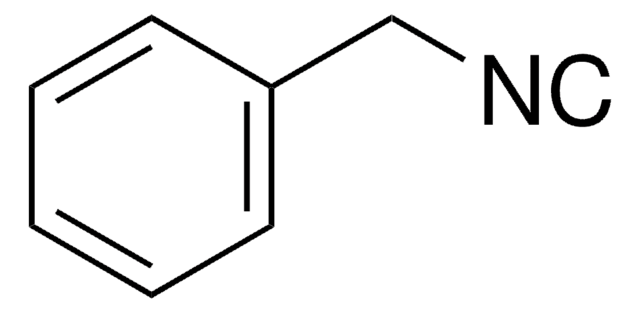
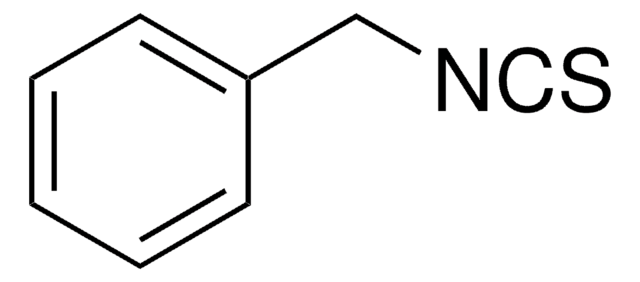
Benzyl isocyanate
99%
Sinónimos:
α-Isocyanatotoluene, (Isocyanatomethyl)benzene, 1-(Isocyanatomethyl)benzene, Phenylmethyl isocyanate
About This Item
Productos recomendados
Nivel de calidad
Ensayo
99%
Formulario
liquid
índice de refracción
n20/D 1.526 (lit.)
bp
101-104 °C/33 mmHg (lit.)
densidad
1.078 g/mL at 25 °C (lit.)
grupo funcional
amine
isocyanate
phenyl
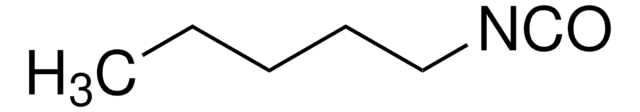
cadena SMILES
O=C=NCc1ccccc1
InChI
1S/C8H7NO/c10-7-9-6-8-4-2-1-3-5-8/h1-5H,6H2
Clave InChI
YDNLNVZZTACNJX-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Descripción general
Aplicación
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Flam. Liq. 3
Código de clase de almacenamiento
3 - Flammable liquids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
113.0 °F - closed cup
Punto de inflamabilidad (°C)
45 °C - closed cup
Equipo de protección personal
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico