701718
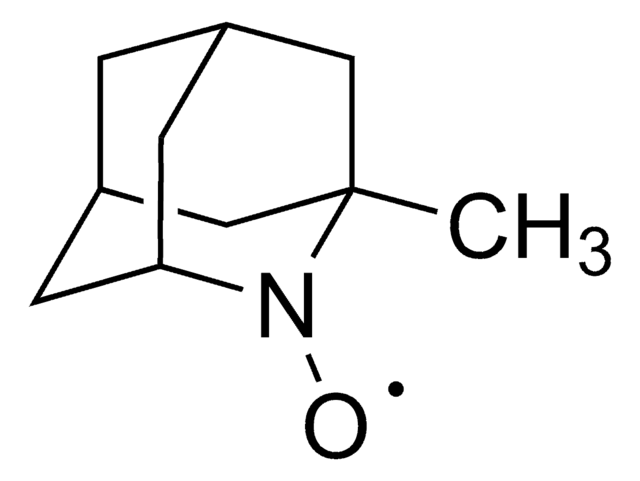
2-Azaadamantan-N-oxyl
90%
Synonym(e):
AZADO
About This Item
Empfohlene Produkte
Assay
90%
Form
powder
Eignung der Reaktion
reagent type: oxidant
mp (Schmelzpunkt)
182-189 °C (D)
Lagertemp.
2-8°C
SMILES String
[O]N1[C@@H]2C[C@H]3C[C@@H](C2)C[C@@H]1C3
InChI
1S/C9H14NO/c11-10-8-2-6-1-7(4-8)5-9(10)3-6/h6-9H,1-5H2/t6-,7+,8-,9+
InChIKey
BCJCJALHNXSXKE-SPJNRGJMSA-N
Verwandte Kategorien
Allgemeine Beschreibung
Anwendung
- As catalyst for the oxidation of wood cellulose.
- As catalyst in the total synthesis of Yaku′amide A, a potential cytotoxin obtained from sponge Ceratopsion sp.
- As oxidant for the oxidation of (S)-glycidol.
Analysenzertifikate (COA)
Suchen Sie nach Analysenzertifikate (COA), indem Sie die Lot-/Chargennummer des Produkts eingeben. Lot- und Chargennummern sind auf dem Produktetikett hinter den Wörtern ‘Lot’ oder ‘Batch’ (Lot oder Charge) zu finden.
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Artikel
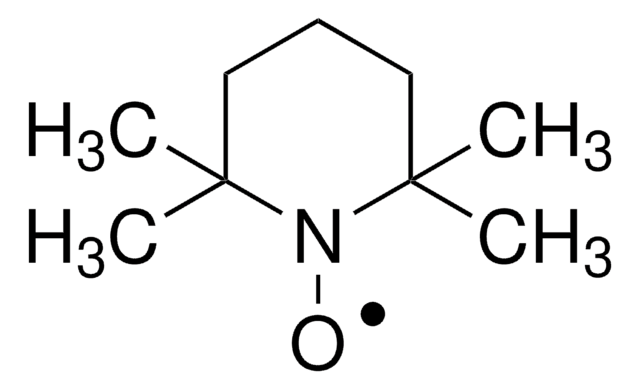
TEMPO (2,2,6,6-Tetramethylpiperidinyloxy or 2,2,6,6-Tetramethylpiperidine 1-oxyl) and its derivatives are stable nitroxy radicals used as catalysts in organic oxidation reactions. TEMPO was discovered by Lebedev and Kazarnovskii in 1960. The stable free radical nature of TEMPO is due to the presence of bulky substituent groups, which hinder the reaction of the free radical with other molecules.
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.![9-Azabicyclo[3.3.1]nonan N-Oxyl 95%](/deepweb/assets/sigmaaldrich/product/structures/287/155/e2f4a2e1-1d4e-4bed-9187-9e16d23cbbbf/640/e2f4a2e1-1d4e-4bed-9187-9e16d23cbbbf.png)