1614669
USP
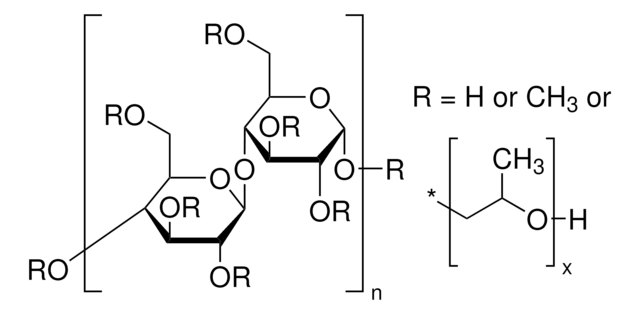
Sodium Starch Glycolate Type A
United States Pharmacopeia (USP) Reference Standard
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Prodotti consigliati
Grado
pharmaceutical primary standard
Famiglia di API
starch
Produttore/marchio commerciale
USP
applicazioni
pharmaceutical (small molecule)
Formato
neat
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Descrizione generale
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the issuing Pharmacopoeia. For further information and support please go to the website of the issuing Pharmacopoeia.
Applicazioni
Sodium Starch Glycolate Type A USP reference standard, intended for use in specified quality tests and assays as specified in the USP compendia. Also, for use with USP monograph such as Sodium Starch Glycolate
Risultati analitici
These products are for test and assay use only. They are not meant for administration to humans or animals and cannot be used to diagnose, treat, or cure diseases of any kind.
Altre note
Sales restrictions may apply.
Prodotti correlati
N° Catalogo
Descrizione
Determinazione del prezzo
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 1
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Certificati d'analisi (COA)
Cerca il Certificati d'analisi (COA) digitando il numero di lotto/batch corrispondente. I numeri di lotto o di batch sono stampati sull'etichetta dei prodotti dopo la parola ‘Lotto’ o ‘Batch’.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
M Maghsoodi et al.
Pharmaceutical development and technology, 16(3), 243-249 (2010-02-24)
Spherical crystallization (SC) of carbamazepine (CBZ) was carried out for preparation of the agglomerates using the solvent change method. The potential of the intraagglomerate addition of sodium starch glycolate (SSG) as a disintegrant agent and povidone (PVP) as a hydrophilic
Aiman A Obaidat et al.
Acta pharmaceutica (Zagreb, Croatia), 61(1), 83-91 (2011-03-17)
The aim of this study was to prepare fast-dissolving tablets of meloxicam after its complexation with β-cyclodextrin (β-CD) and to investigate the effect of using different superdisintegrants on the disintegration and release of meloxicam from the tablets. A complex of
Sagarika Bose et al.
International journal of pharmaceutics, 393(1-2), 32-40 (2010-02-09)
Film coating is generally accomplished by spraying polymers dissolved in solvents onto a cascading bed of tablets. The limitations associated with the use of solvents (both aqueous and organic) can be overcome by the use of solventless coating technologies. In
Balasubramaniam Jagadish et al.
Chemical & pharmaceutical bulletin, 58(3), 293-300 (2010-03-02)
The present study investigated the effect of co-grinding raloxifene HCL (RHCL) with different superdisintegrants, namely crospovidone (CP), croscarmellose sodium (CCS) and sodium starch glycolate (SSG), using a ball mill, in order to determine the potential effect on dissolution rate and
Ramji Anil Kumar Arza et al.
AAPS PharmSciTech, 10(1), 220-226 (2009-03-12)
Drugs that have narrow absorption window in the gastrointestinal tract (GIT) will have poor absorption. For these drugs, gastroretentive drug delivery systems offer the advantage in prolonging the gastric emptying time. Swellable, floating, and sustained release tablets are developed by
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.