SRE0045
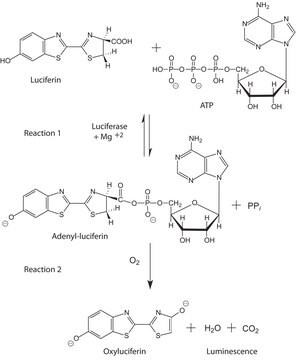
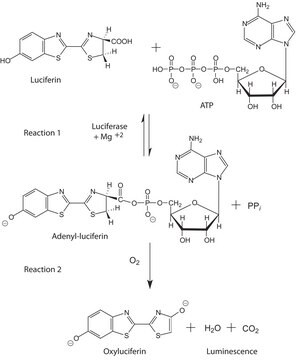
Luciferase from Photinus pyralis (firefly)
recombinant, expressed in E. coli, lyophilized powder, ≥10×1010 units/mg protein
Sinonimo/i:
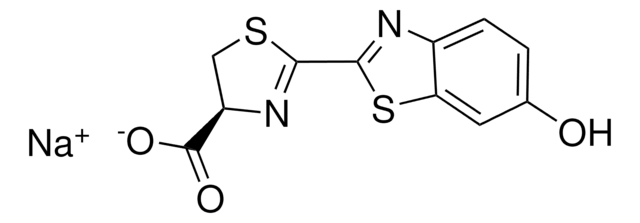
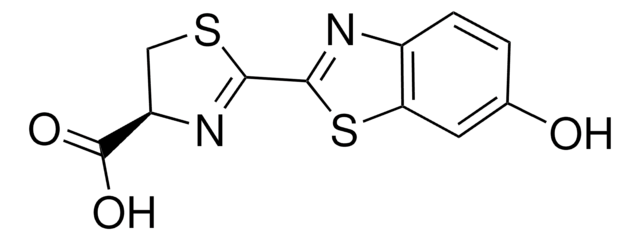
Luciferase firefly
About This Item
Prodotti consigliati
Ricombinante
expressed in E. coli
Livello qualitativo
Forma fisica
lyophilized powder
Attività specifica
≥10×1010 units/mg protein
PM
62 kDa
applicazioni
diagnostic assay manufacturing
Temperatura di conservazione
−20°C
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Applicazioni
Azioni biochim/fisiol
Definizione di unità
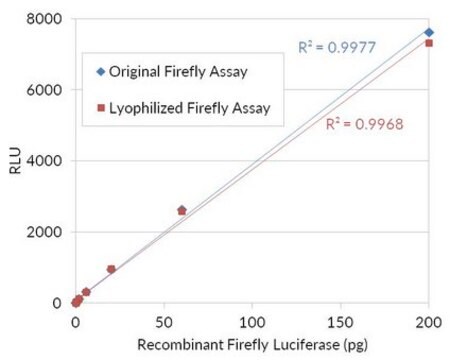
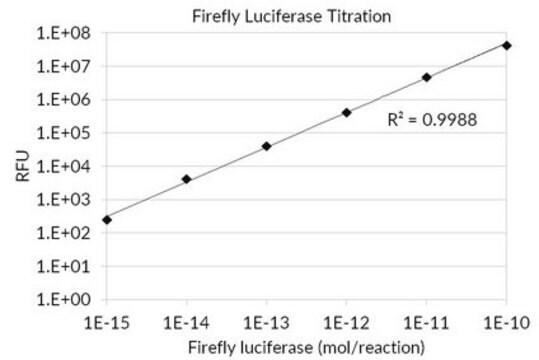
Unit Definition Conversion Factor: There are approximately 9000 Relative Light Units (RLU) per one traditional Light Unit that uses a peak height equivalent to 0.02 μCi of 14C in a PPO/POPOP cocktail.
Stato fisico
Nota sulla preparazione
After reconstitution, the enzyme solutions can kept at 4-8 °C for up to 2 days or frozen in working aliquots at -20°C for at least one month. Repeated freezing and thawing is not recommended.
Substrato
Avvertenze
Danger
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Eye Dam. 1 - Resp. Sens. 1
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Certificati d'analisi (COA)
Cerca il Certificati d'analisi (COA) digitando il numero di lotto/batch corrispondente. I numeri di lotto o di batch sono stampati sull'etichetta dei prodotti dopo la parola ‘Lotto’ o ‘Batch’.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Articoli
Low cost and high performing bioluminescent Firefly Luciferase and Renilla Luciferase assays for gene reporter and ATP cell viability experiments. Benefits of dual luciferase assays also discussed.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.