M5255
Mycophenolic acid
from Penicillium brevicompactum, ≥98% (HPLC), powder, IMP dehydrogenase inhibitor
Sinonimo/i:
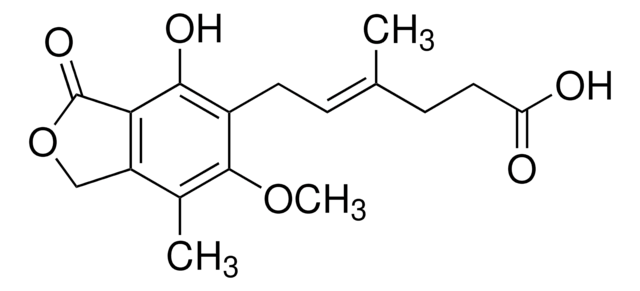
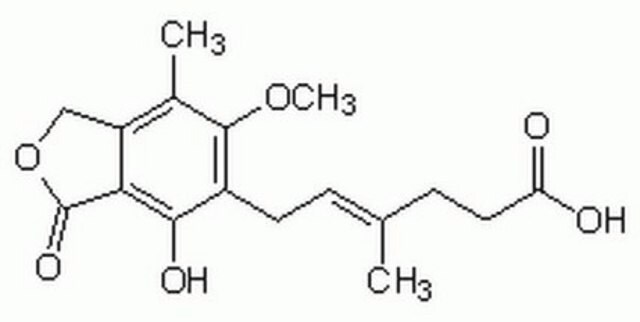
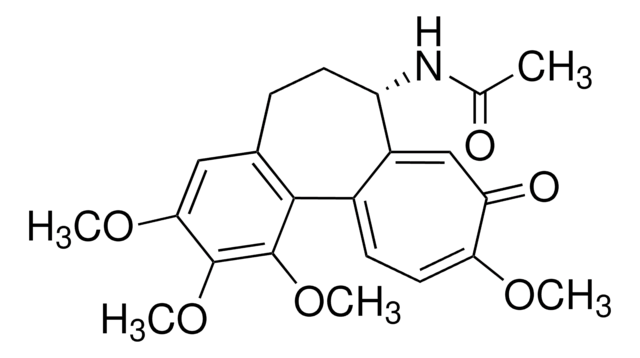
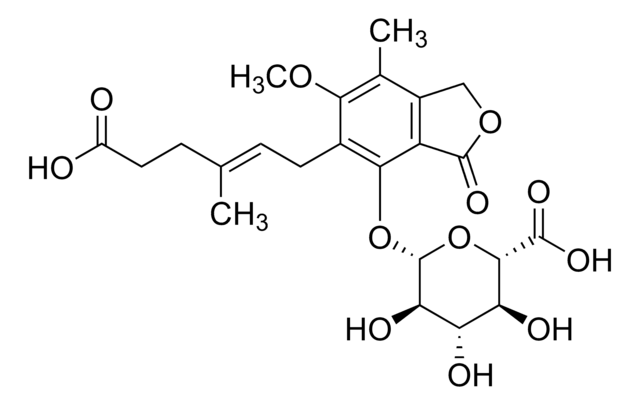
6-(1,3-Dihydro-7-hydroxy-5-methoxy-4-methyl-1-oxoisobenzofuran-6-yl)-4-methyl-4-hexanoic acid, 6-(4-Hydroxy-6-methoxy-7-methyl-3-oxo-5-phthalanyl)-4-methyl-4-hexenoic acid, NSC 129185
About This Item
Prodotti consigliati
Nome del prodotto
Mycophenolic acid, ≥98%
Origine biologica
Penicillium brevicompactum
Livello qualitativo
Saggio
≥98%
Colore
white to yellow-white
Punto di fusione
<143.0 °C
Solubilità
methanol: 49.00-51.00 mg/mL, clear, colorless to faintly yellow
Modalità d’azione
enzyme | inhibits
Ideatore
Novartis
Temperatura di conservazione
2-8°C
Stringa SMILE
COc1c(C)c2COC(=O)c2c(O)c1C\C=C(/C)CCC(O)=O
InChI
1S/C17H20O6/c1-9(5-7-13(18)19)4-6-11-15(20)14-12(8-23-17(14)21)10(2)16(11)22-3/h4,20H,5-8H2,1-3H3,(H,18,19)/b9-4+
HPNSFSBZBAHARI-RUDMXATFSA-N
Informazioni sul gene
human ... IMPDH1(3614) , IMPDH2(3615)
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Applicazioni
Azioni biochim/fisiol
Caratteristiche e vantaggi
Avvertenza
Nota sulla preparazione
Avvertenze
Danger
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Muta. 2 - Repr. 1B - STOT RE 1 Oral
Organi bersaglio
Immune system
Codice della classe di stoccaggio
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Articoli
Neoplastic cells are highly dependent on the de novo synthesis of nucleotides to maintain sufficient pools to support DNA replication and the production of RNA.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.