G6782
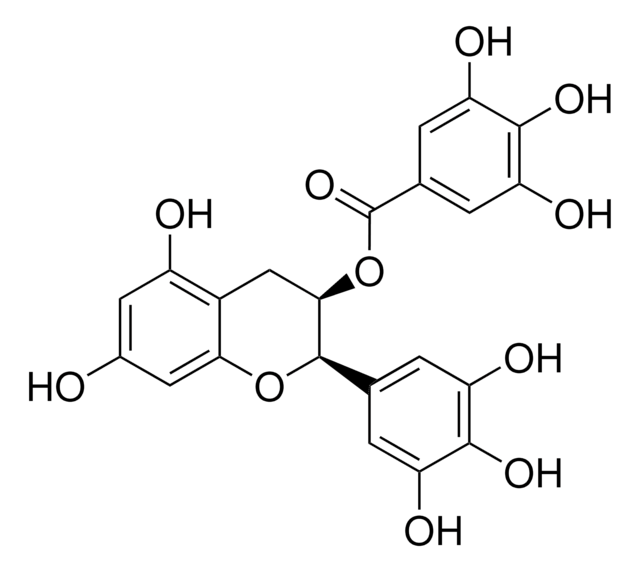
(−)-Gallocatechin gallate
from green tea, ≥98% (HPLC)
Sinonimo/i:
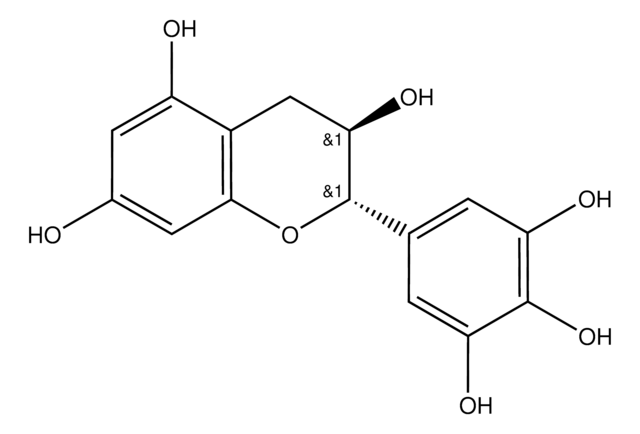
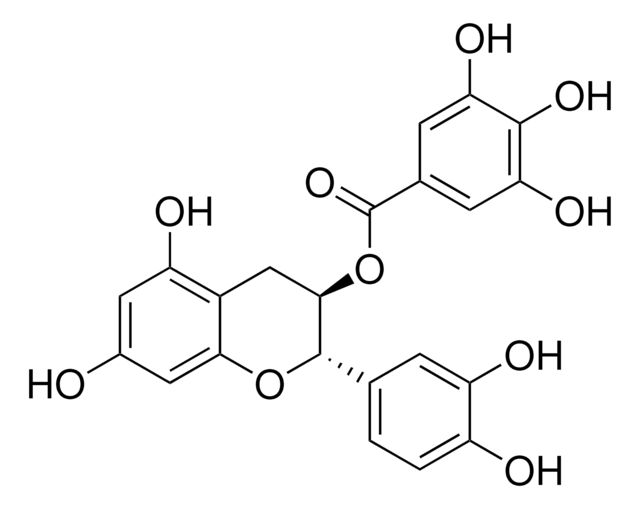
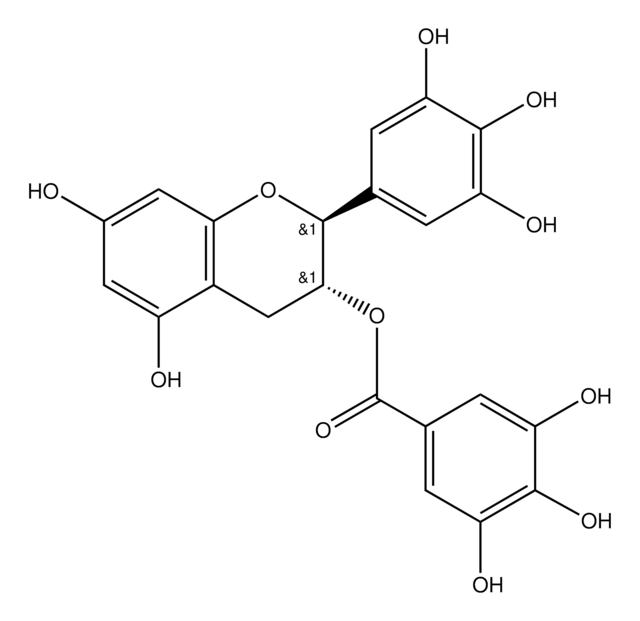
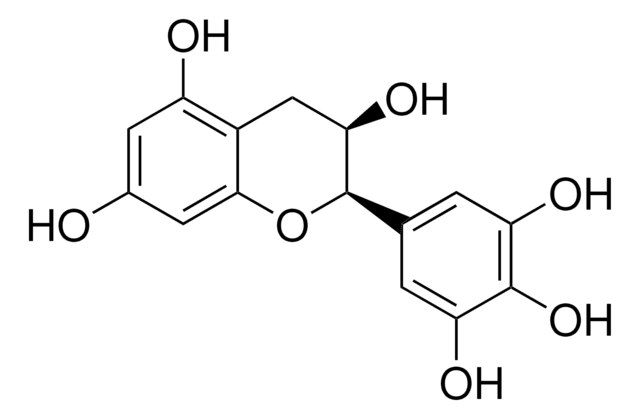
(2S,3R)-2-(3,4,5-Trihydroxyphenyl)-3,4-dihydro-1(2H)-benzopyran-3,5,7-triol 3-(3,4,5-trihydroxybenzoate)
About This Item
Prodotti consigliati
Origine biologica
green tea
Livello qualitativo
Saggio
≥98% (HPLC)
Stato
powder
applicazioni
metabolomics
vitamins, nutraceuticals, and natural products
Temperatura di conservazione
2-8°C
Stringa SMILE
Oc1cc(O)c2C[C@@H](OC(=O)c3cc(O)c(O)c(O)c3)[C@@H](Oc2c1)c4cc(O)c(O)c(O)c4
InChI
1S/C22H18O11/c23-10-5-12(24)11-7-18(33-22(31)9-3-15(27)20(30)16(28)4-9)21(32-17(11)6-10)8-1-13(25)19(29)14(26)2-8/h1-6,18,21,23-30H,7H2/t18-,21+/m1/s1
WMBWREPUVVBILR-NQIIRXRSSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Descrizione generale
Applicazioni
- to perform the tannase activity assay of the recombinant Lactobacillus plantarum tannase enzyme
- to study its inhibitory effects on HIV-1 integrase activity
- to study its regulatory mechanism on cholesterol metabolism in rat liver
- to analyze its effect as a polyphenol on human spermatozoa and their application in assisted reproductive technology
- to study the interference in stress protective activity of cellular prion protein (PrPC) and the formation of scrapie prion protein (PrPSc)
Azioni biochim/fisiol
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organi bersaglio
Respiratory system
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Articoli
Antioxidants protect biological systems from oxidative damage produced by oxygen-containing free radicals and from redoxactive transition metal ions such as iron, copper, and cadmium.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.