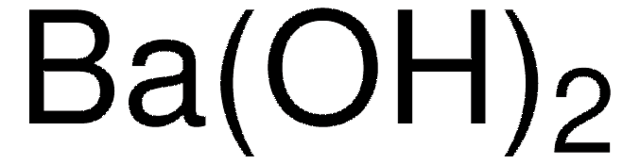B4059
Barium hydroxide solution
0.3 N
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
Ba(OH)2
Numero CAS:
Peso molecolare:
171.34
Numero MDL:
Codice UNSPSC:
12352303
ID PubChem:
NACRES:
NA.25
Prodotti consigliati
Stato
liquid
Livello qualitativo
Concentrazione
0.3 N
Solubilità
soluble (Clear colorless liquid, may contain some small insoluble particles.)
Stringa SMILE
O[Ba]O
InChI
1S/Ba.2H2O/h;2*1H2/q+2;;/p-2
RQPZNWPYLFFXCP-UHFFFAOYSA-L
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Categorie correlate
Applicazioni
Barium hydroxide may be used in the development of formulations for the purification of materials such as heteropolysaccharide-protein biopolymers and the release of long-chain bases (LCB) from biological samples. Barium hydroxide solution may be used for protein precipitation in turbid fluids such as whole blood in assays based on the Somogyi-Nelson method.
Used in conjunction with zinc sulfate to deproteinize via barium sulfate precipitation whole blood, plasma, or serum samples which are colored or turbid. The resulting supernatant or filtrate is then available for subsequent analysis of glucose by Somogi-Nelson method.
Prodotti correlati
N° Catalogo
Descrizione
Determinazione del prezzo
Avvertenze
Danger
Indicazioni di pericolo
Classi di pericolo
Eye Dam. 1 - Skin Corr. 1
Codice della classe di stoccaggio
8B - Non-combustible corrosive hazardous materials
Classe di pericolosità dell'acqua (WGK)
WGK 1
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Bahareh Tabatabaee Amid et al.
Molecules (Basel, Switzerland), 17(9), 10875-10892 (2012-09-12)
Natural biopolymers from plant sources contain many impurities (e.g., fat, protein, fiber, natural pigment and endogenous enzymes), therefore, an efficient purification process is recommended to minimize these impurities and consequently improve the functional properties of the biopolymer. The main objective
E Bach et al.
Analytical biochemistry, 203(2), 335-339 (1992-06-01)
A rapid, facile, and sensitive uv-spectrophotometric assay has been developed for the determination of the enzymatic degradation of polysaccharides that generates reducing sugars. The assay was carried out with 2-cyanoacetamide in a single test tube. The solution was left at
M F Byford
The Biochemical journal, 280 ( Pt 1), 261-265 (1991-11-15)
The beta-elimination of phosphoserine residues by dilute alkali is catalysed by the presence of group II metal ions. The use of 0.1 M-Ba (OH)2 catalysed the rate of beta-elimination of phosphoserine by more than two orders of magnitude compared with
Spontaneous ignition, explosion, and fire with sevoflurane and barium hydroxide lime.
Junzheng Wu et al.
Anesthesiology, 101(2), 534-537 (2004-07-28)
Marshall B Dunning et al.
Anesthesiology, 106(1), 144-148 (2007-01-02)
Fires, explosions, and extreme heat production may occur when sevoflurane reacts with desiccated barium hydroxide lime. The identity of the flammable gas has not previously been published, although carbon monoxide, methanol, formaldehyde, and methyl formate have been identified in low
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.