94335
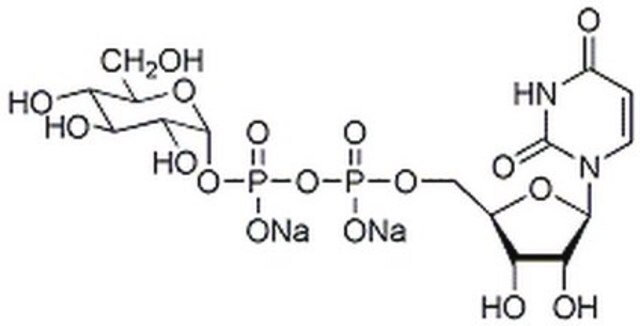
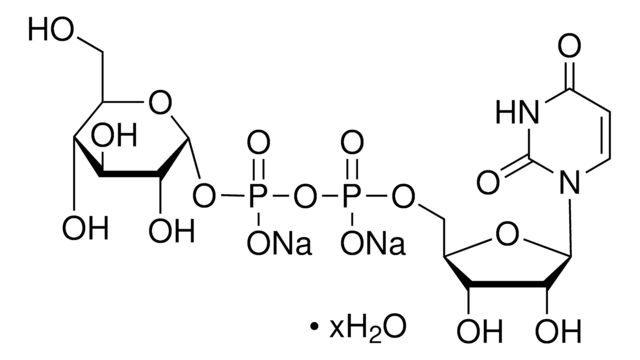
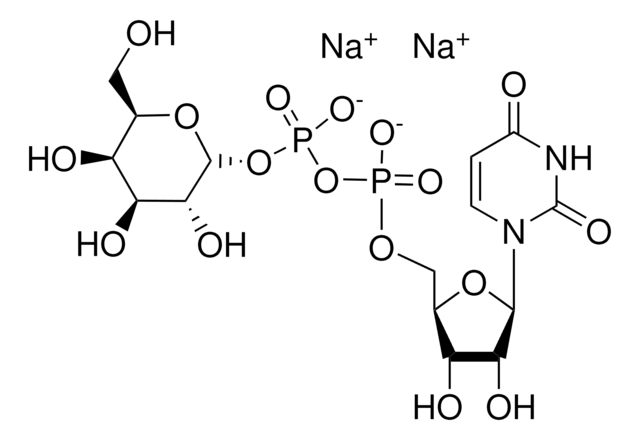
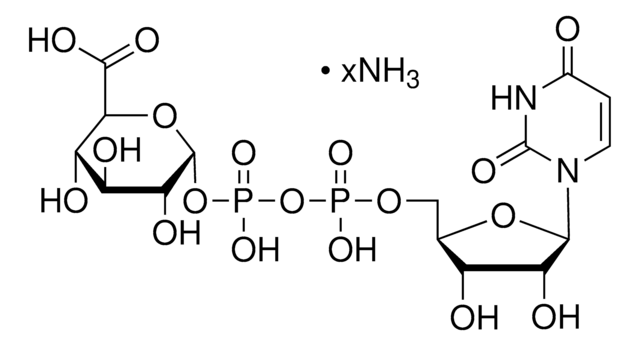
Uridine 5′-diphosphoglucose disodium salt
≥98.0% (HPLC)
Sinonimo/i:
UDP-D-Glucose disodium salt, UDPG, UDP-glc, Uridine-diphosphate-glucose disodium salt, Uridine[5’]diphospho[1]-α-D-glucopyranose disodium salt
About This Item
Prodotti consigliati
Origine biologica
Saccharomyces cerevisiae
Saggio
≥98.0% (HPLC)
Stato
powder
Impurezze
≤5% solvent
≤8.5% water
Solubilità
H2O: 50 mg/mL, clear
Temperatura di conservazione
−20°C
Stringa SMILE
[Na+].[Na+].OC[C@H]1O[C@H](OP([O-])(=O)OP([O-])(=O)OC[C@H]2O[C@H]([C@H](O)[C@@H]2O)N3C=CC(=O)NC3=O)[C@H](O)[C@@H](O)[C@@H]1O
InChI
1S/C15H24N2O17P2.2Na/c18-3-5-8(20)10(22)12(24)14(32-5)33-36(28,29)34-35(26,27)30-4-6-9(21)11(23)13(31-6)17-2-1-7(19)16-15(17)25;;/h1-2,5-6,8-14,18,20-24H,3-4H2,(H,26,27)(H,28,29)(H,16,19,25);;/q;2*+1/p-2/t5-,6-,8-,9-,10+,11-,12-,13-,14-;;/m1../s1
PKJQEQVCYGYYMM-QBNUFUENSA-L
Descrizione generale
Applicazioni
- as a substrate in the enzymatic production of glycosides and their detection by liquid chromatography-mass spectrometry.
- as a substrate in the substrate screening and binding affinity measurements of the human CMP-Sia transporter.
Azioni biochim/fisiol
Altre note
Sostituito da
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.