G2164
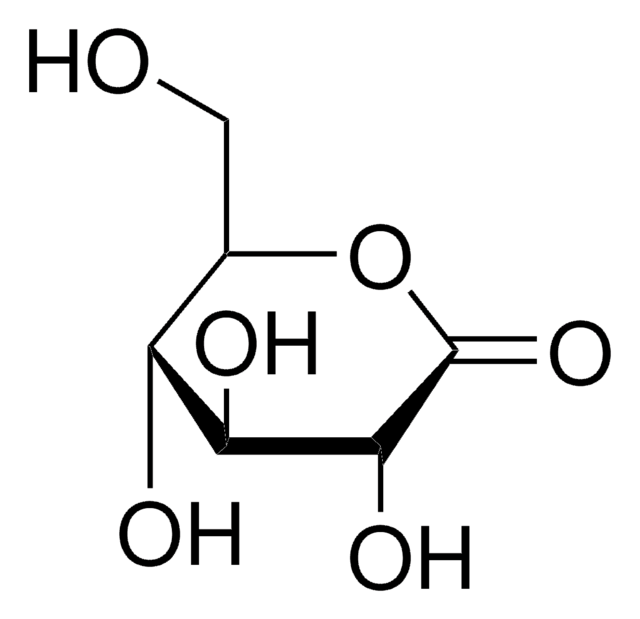
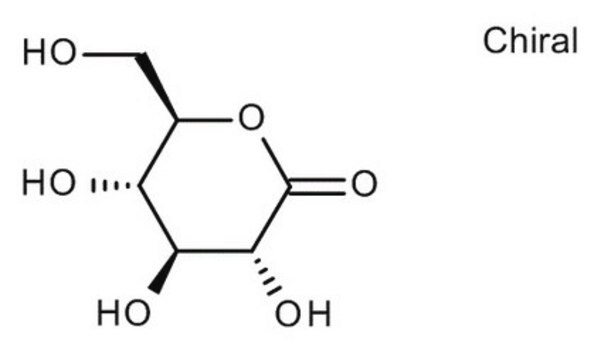
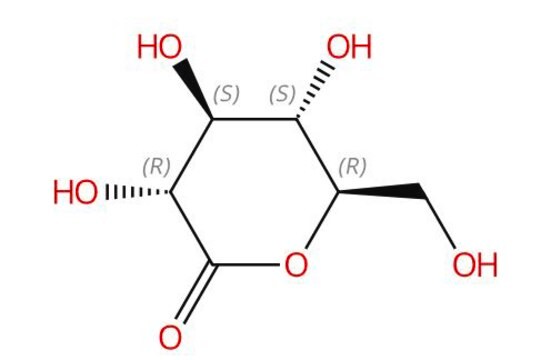
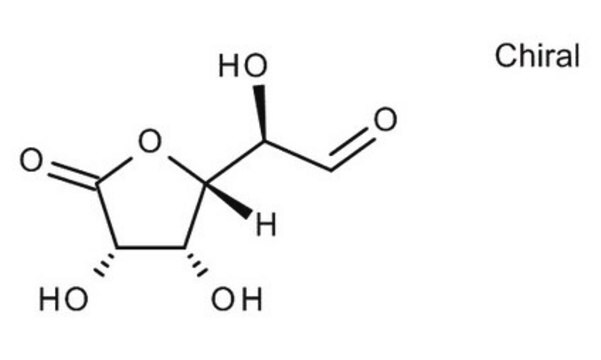
Gluconolactone
99.0-101.0%, meets USP testing specifications
Sinonimo/i:
D-(+)-Gluconic acid δ-lactone, δ-Gluconolactone, 1,2,3,4,5-Pentahydroxycaproic acid δ-lactone, D-(+)-Dextronic acid δ-lactone
About This Item
Prodotti consigliati
Origine biologica
corn
Livello qualitativo
agenzia
USP/NF
meets USP testing specifications
Saggio
99.0-101.0%
Forma fisica
crystalline
Colore
white
Punto di fusione
160 °C (dec.) (lit.)
Solubilità
water: soluble
applicazioni
pharmaceutical (small molecule)
Stringa SMILE
OC[C@H]1OC(=O)[C@H](O)[C@@H](O)[C@@H]1O
InChI
1S/C6H10O6/c7-1-2-3(8)4(9)5(10)6(11)12-2/h2-5,7-10H,1H2/t2-,3-,4+,5-/m1/s1
PHOQVHQSTUBQQK-SQOUGZDYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Categorie correlate
Descrizione generale
Applicazioni
- to control the acidification kinetics of natural rubber latex (NRL) suspensions
- to study its effects on inhibition and disintegration of collagen fibrils
- to prepare alginate hydrogel
Azioni biochim/fisiol
Altre note
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 1
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.