5.33978
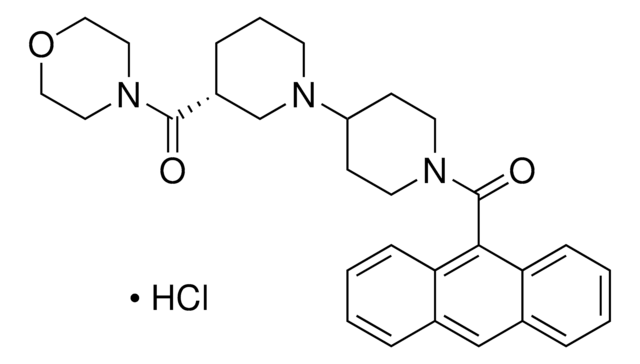
ACC Inhibitor IV, CP-640186
Sinonimo/i:
ACC Inhibitor IV, CP-640186, 9-Anthryl((3 R)-3-(4-morpholinylcarbonyl)-1,4ʹ-bipiperidin-1ʹ-yl)methanone, HCl, Acetyl-CoA Carboxylase Inhibitor IV, CP-640186, HCl
About This Item
Prodotti consigliati
Saggio
≥98% (HPLC)
Livello qualitativo
Forma fisica
solid
Produttore/marchio commerciale
Calbiochem®
Condizioni di stoccaggio
OK to freeze
desiccated (hygroscopic)
protect from light
Colore
light orange
Solubilità
DMSO: 50 mg/mL
H2O: 50 mg/mL
Temperatura di conservazione
2-8°C
Descrizione generale
Please note that the molecular weight for this compound is batch-specific due to variable water content. Please refer to the vial label or the certificate of analysis for the batch-specific molecular weight. The molecular weight provided represents the baseline molecular weight without water.
Azioni biochim/fisiol
Acetyl-CoA carboxylase (ACC)
Attenzione
Stato fisico
Ricostituzione
Altre note
Gu, Y.G., et al. 2006. J. Med. Chem.49, 3770.
Zhang, H., et al. 2004. Structure12, 1683.
Harwood, H.J., et al. 2003. J. Biol. Chem.278, 37099.
Note legali
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Certificati d'analisi (COA)
Cerca il Certificati d'analisi (COA) digitando il numero di lotto/batch corrispondente. I numeri di lotto o di batch sono stampati sull'etichetta dei prodotti dopo la parola ‘Lotto’ o ‘Batch’.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.