K3007
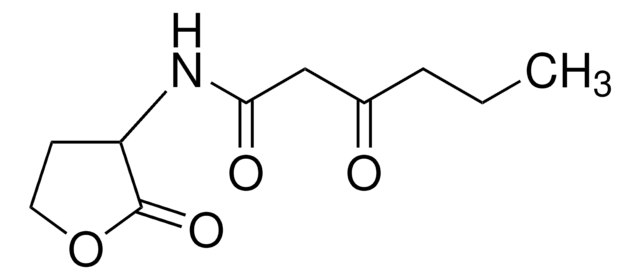
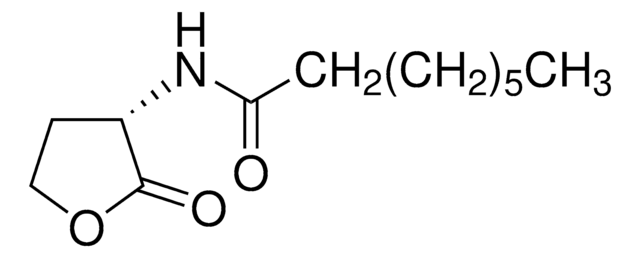
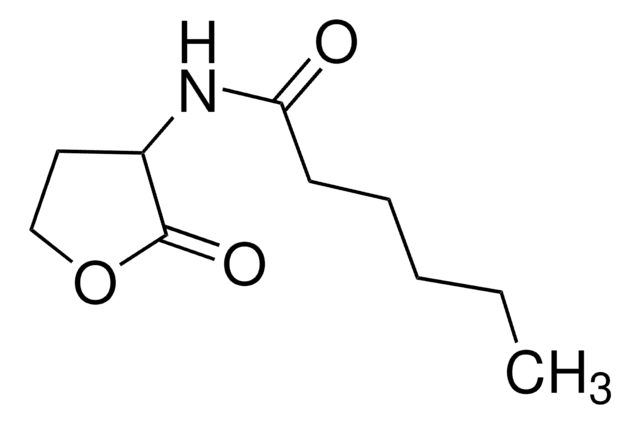
N-(β-Ketocaproyl)-L-homoserine lactone
≥98%, detection, for peptide synthesis
Sinonimo/i:
N-(3-Oxohexanoyl)-L-homoserine lactone
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula empirica (notazione di Hill):
C10H15NO4
Numero CAS:
Peso molecolare:
213.23
Numero MDL:
Codice UNSPSC:
12352209
eCl@ss:
32160406
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Nome del prodotto
N-(β-Ketocaproyl)-L-homoserine lactone, ≥98%
Saggio
≥98%
Stato
powder
Colore
white
applicazioni
detection
peptide synthesis
Temperatura di conservazione
−20°C
Stringa SMILE
CCCC(=O)CC(=O)N[C@H]1CCOC1=O
InChI
1S/C10H15NO4/c1-2-3-7(12)6-9(13)11-8-4-5-15-10(8)14/h8H,2-6H2,1H3,(H,11,13)/t8-/m0/s1
YRYOXRMDHALAFL-QMMMGPOBSA-N
Descrizione generale
N-(β-Ketocaproyl)-L-homoserine lactone (3-oxo-C6-HSL) is one of the quorum signaling molecules for V. fischeri. 3-oxo-C6-HSL is produced by LuxI autoinducer synthase catalyzed reaction between S-adenosylmethionine and acylated-acyl carrier proteins (Acyl-ACPs). It can be used in analyzing the expression of the orthogonal specialized reporter genes.
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Xiaofei Qin et al.
Biomolecules, 8(3) (2018-09-06)
New approaches to deal with drug-resistant pathogenic bacteria are urgent. We studied the antibacterial effect of chitosans against an Escherichia coli quorum sensing biosensor reporter strain and selected a non-toxic chitosan to evaluate its quorum sensing (QS) inhibition activity and
Nikolay A Aleksashin et al.
Nature communications, 11(1), 1858-1858 (2020-04-22)
Ribosome engineering is a powerful approach for expanding the catalytic potential of the protein synthesis apparatus. Due to the potential detriment the properties of the engineered ribosome may have on the cell, the designer ribosome needs to be functionally isolated from the
John T Sexton et al.
Molecular systems biology, 16(7), e9618-e9618 (2020-07-17)
The engineering of advanced multicellular behaviors, such as the programmed growth of biofilms or tissues, requires cells to communicate multiple aspects of physiological information. Unfortunately, few cell-cell communication systems have been developed for synthetic biology. Here, we engineer a genetically
Rong Tang et al.
International journal of food microbiology, 297, 60-71 (2019-03-19)
Quorum sensing (QS) is crucial for adaption and development of foodborne bacteria in diverse environments. Pseudomonas fluorescens PF07 with QS mediated acylated homoserine lactones (AHLs) activity was isolated from spoiled large yellow croaker (Pseudosciaena crocea). In this study AHL-mediated QS
Ling Yan et al.
Journal of microbiological methods, 68(1), 40-45 (2006-08-19)
The autoinducer N-(3-oxo-hexanoyl)-L-homoserine lactone (3-oxo-C6-HSL) plays a significant role in the quorum-sensing system of the marine bacterium Vibrio fischeri. Upon forming a transcriptional activation complex with LuxR, 3-oxo-C6-HSL induces transcription of the luxICDABEG operon, leading to the increased production of
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.