D57558
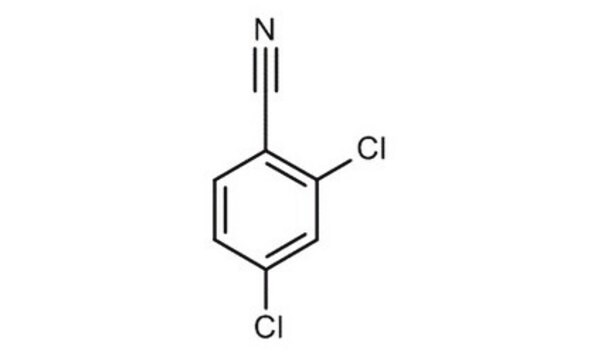
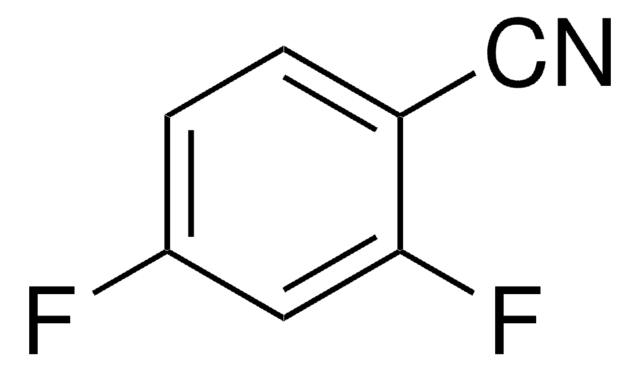
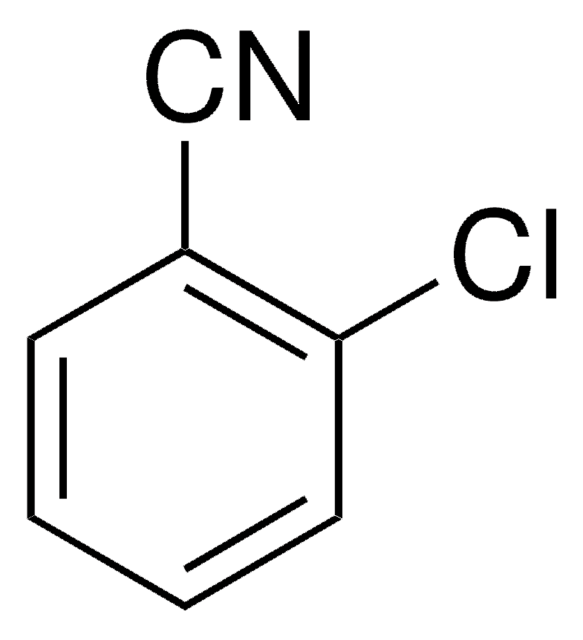
2,6-Dichlorobenzonitrile
97%
Sinonimo/i:
Dichlobenil
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
Cl2C6H3CN
Numero CAS:
Peso molecolare:
172.01
Beilstein:
1909167
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
97%
Stato
powder
Punto di fusione
143-146 °C (lit.)
Stringa SMILE
Clc1cccc(Cl)c1C#N
InChI
1S/C7H3Cl2N/c8-6-2-1-3-7(9)5(6)4-10/h1-3H
YOYAIZYFCNQIRF-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Applicazioni
2,6-Dichlorobenzonitrile can be used as a starting material to synthesize:
- 2,6-Dichlorobenzaldehyde using lithium N, N′-dimethylethylenediaminoaluminum hydride as a reducing agent.
- 5-(2,6-Dichlorophenyl)-2H-tetrazole via gold-catalyzed nucleophilic (3 + 2) cycloaddition reaction with sodium azide.
- 2,6-Dichlorobenzamide via hydrolysis using potassium tert-butoxide as a catalyst.
- Chloro-aminoindazole by reacting with hydrazine monohydrate.
- 2,6-Dichlorobenzenecarboselenoamide by treating with Woollins′ reagent.
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Acute Tox. 4 Dermal - Aquatic Chronic 2
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 2
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Synthesis of primary arylselenoamides by reaction of aryl nitriles with Woollins' reagent
Hua Guoxiong, et al.
Organic Letters, 8(23), 5251-5254 (2006)
Valeria Franceschini et al.
Stem cells (Dayton, Ohio), 27(4), 825-835 (2009-04-08)
The herbicide dichlobenil selectively causes necrosis of the dorsomedial part of olfactory neuroepithelium (NE) with permanent damage to the underlying mucosa, whereas the lateral part of the olfactory region and the nasal respiratory mucosa remain undamaged. We investigated here whether
L Peng et al.
Plant biology (Stuttgart, Germany), 15(2), 405-414 (2012-07-05)
Cellulose is the major component of plant cell walls and is an important source of industrial raw material. Although cellulose biosynthesis is one of the most important biochemical processes in plant biology, the regulatory mechanisms of cellulose synthesis are still
Fang Xie et al.
Toxicology and applied pharmacology, 272(3), 598-607 (2013-08-08)
We explored the mechanisms underlying the differential effects of two olfactory toxicants, the herbicide 2,6-dichlorobenzonitrile (DCBN) and the anti-thyroid drug methimazole (MMZ), on olfactory receptor neuron (ORN) regeneration in mouse olfactory epithelium (OE). DCBN, but not MMZ, induced inflammation-like pathological
Guang-Cai Chen et al.
Journal of hazardous materials, 188(1-3), 156-163 (2011-02-18)
The effect of lead on the adsorption of diuron and dichlobenil on multiwalled carbon nanotubes (MWCNTs) was investigated to explore the possible application of MWCNTs for removal of both herbicides from contaminated water. The adsorption of diuron and dichlobenil on
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.