747157
Potassium pentafluroroethyltrifluoroborate
95%
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
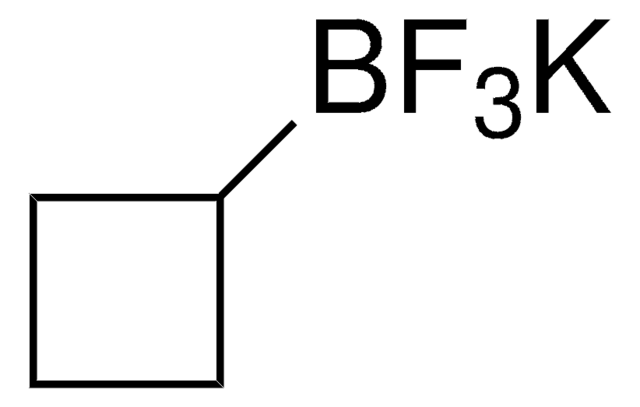
Formula empirica (notazione di Hill):
C2BF8K
Numero CAS:
Peso molecolare:
225.92
Numero MDL:
Codice UNSPSC:
12352103
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
95%
Stato
solid
Punto di fusione
252-257 °C
Gruppo funzionale
fluoro
Temperatura di conservazione
2-8°C
Stringa SMILE
F[B-](F)(F)C(F)(F)C(F)(F)F.[K+]
InChI
1S/C2BF8.K/c4-1(5,2(6,7)8)3(9,10)11;/q-1;+1
PSJPJAFBTMLFFX-UHFFFAOYSA-N
Categorie correlate
Applicazioni
Organotrifluoroborate involved in:
Organotrifluoroborates as versatile and stable boronic acid surrogates
- Suzuki Miyaura cross-coupling reactions, and polymerization reactions
- Synthesis of photonic crystals
- Synthesis of sensitizers for dye-sensitized solar cells
- Mannich / diastereoselective hydroamination reaction sequence
Organotrifluoroborates as versatile and stable boronic acid surrogates
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organi bersaglio
Respiratory system
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Scegli una delle versioni più recenti:
Certificati d'analisi (COA)
Lot/Batch Number
Non trovi la versione di tuo interesse?
Se hai bisogno di una versione specifica, puoi cercare il certificato tramite il numero di lotto.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Gary A Molander et al.
Journal of the American Chemical Society, 130(47), 15792-15793 (2008-11-05)
A method was developed for the hydroboration of alkenyl-containing organotrifluoroborates to generate dibora intermediates. The reactivity differences between organotrifluoroborates and trialkylboranes facilitated the cross-coupling of the borane moiety of these intermediates in a highly chemoselective fashion with aryl halides, leaving
Roberto Grisorio et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 16(27), 8054-8061 (2010-06-04)
The mechanism of the Suzuki-Heck (SuHe) polymerisation of 2,7-dibromo-9,9-di(n-dodecyl)fluorene (1) with potassium vinyl trifluoroborate (PVTB) for the synthesis of poly(fluorenylene vinylene)s (PFVs) has been investigated. In the first stage, a palladium-catalysed chain-growth AA/B(C)-type polycondensation occurs, as evidenced by the linear
Emilio Alacid et al.
The Journal of organic chemistry, 74(6), 2321-2327 (2009-02-17)
Potassium vinyl and alkenyltrifluoroborates are cross-coupled with aryl and heteroaryl bromides using 1 mol % Pd loading of 4-hydroxyacetophenone oxime derived palladacycle or Pd(OAc)2 as precatalysts, K2CO3 as base, and TBAB as additive and water reflux under conventional or microwave
S. Achelle;
Journal of Polymer Science: Part A, General Papers, 48, 2659-2665 (2010)
Jenny M Baxter Vu et al.
Organic letters, 13(15), 4056-4059 (2011-07-14)
A new two-step synthesis of highly substituted pyrrolidines has been developed. Chiral silane Lewis acid promoted enantioselective Mannich reactions of silyl ketene imines with acylhydrazones may be used to access bishomoallylic benzoic hydrazides that in turn may be cyclized to
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.
![Phenyl[3-(trifluoromethyl)phenyl]iodonium triflate ≥98% (HPLC)](/deepweb/assets/sigmaaldrich/product/structures/424/062/057593f4-e032-4d3e-bed6-6be37c1ae76d/640/057593f4-e032-4d3e-bed6-6be37c1ae76d.png)