522856
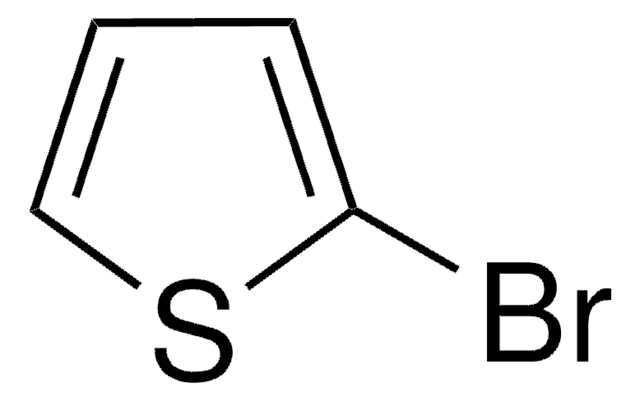
5-Bromo-2,2′-bithiophene
96%
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula empirica (notazione di Hill):
C8H5BrS2
Numero CAS:
Peso molecolare:
245.16
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
96%
Stato
solid
Punto di fusione
29-32 °C (lit.)
Stringa SMILE
Brc1ccc(s1)-c2cccs2
InChI
1S/C8H5BrS2/c9-8-4-3-7(11-8)6-2-1-5-10-6/h1-5H
OMOAIGVIYUXYAU-UHFFFAOYSA-N
Descrizione generale
5-Bromo-2,2′-bithiophene is a bromothiophene derivative. Its reaction with various aryl iodides bearing an electron-donating or electron-withdrawing substituent has been described. It can be synthesized from 2,2′-bithiophene.
Applicazioni
5-Bromo-2,2′-bithiophene may be used in the synthesis of trimethyl-[2,2′;5′,2″;5″,2″]quaterthiophen-5-yl-silane (4TTMS) and 5-hexylsulfanyl-2,2′:5′,2′′-terthiophene.
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Mark E Roberts et al.
Proceedings of the National Academy of Sciences of the United States of America, 105(34), 12134-12139 (2008-08-20)
The development of low-cost, reliable sensors will rely on devices capable of converting an analyte binding event to an easily read electrical signal. Organic thin-film transistors (OTFTs) are ideal for inexpensive, single-use chemical or biological sensors because of their compatibility
Kei Kobayashi et al.
Organic letters, 7(22), 5083-5085 (2005-10-21)
[reaction: see text] Bromothiophene derivatives react with aryl iodides catalyzed by a palladium complex in the presence of a silver(I) nitrate/potassium fluoride system to induce coupling at the C-H bond, while the carbon-bromine bond is intact. The produced coupling product
Silole-Containing. pi.-Conjugated Systems. 3.1 A Series of Silole-Thiophene Cooligomers and Copolymers: Synthesis, Properties, and Electronic Structures.
Tamao K, et al.
Macromolecules, 28(25), 8668-8675 (1995)
Three-dimensional tetra (oligothienyl) silanes as donor material for organic solar cells.
Roquet S, et al.
Journal of Materials Chemistry, 16(29), 3040-3045 (2006)
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.

![Benzo[1,2-b:4,5-b′]dithiophene-4,8-dione 97%](/deepweb/assets/sigmaaldrich/product/structures/418/544/b7faac0b-ad09-4b42-a9fa-aeb38017a39e/640/b7faac0b-ad09-4b42-a9fa-aeb38017a39e.png)
![Thieno[3,2-b]thiophene 95%](/deepweb/assets/sigmaaldrich/product/structures/353/609/429fd4bf-e217-4371-80a3-9e5a4d88908b/640/429fd4bf-e217-4371-80a3-9e5a4d88908b.png)
![2,5-Bis(trimethylstannyl)-thieno[3,2-b]thiophene 97%](/deepweb/assets/sigmaaldrich/product/structures/126/532/26557e94-858e-4c96-90de-ca88d84a8727/640/26557e94-858e-4c96-90de-ca88d84a8727.png)