457698
(R)-(+)-2-Methyl-CBS-oxazaborolidine solution
1 M in toluene
Sinonimo/i:
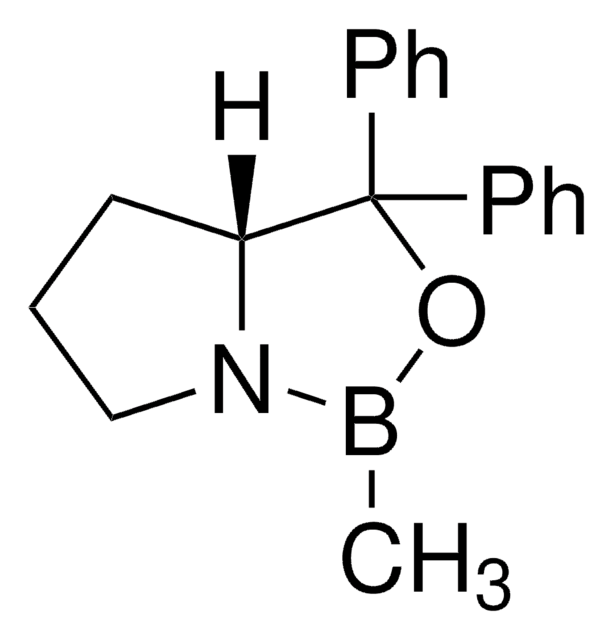
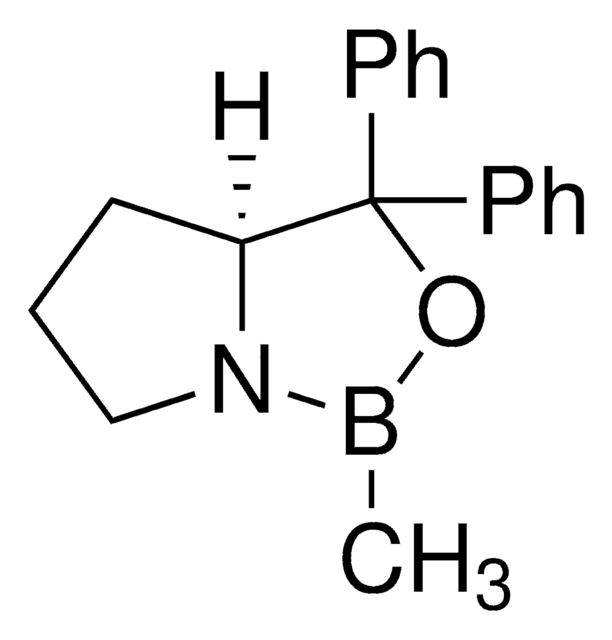
α,α-Diphenyl-D-prolinolmethylboronic acid cyclamide ester, (R)-1-Methyl,3,3-diphenyl-tetrahydro-pyrrolo(1,2-c)(1,3,2)oxazaborole, (R)-Tetrahydro-1-methyl-3,3-diphenyl-1H,3H-pyrrolo[1,2-c][1,3,2]oxazaborole
About This Item
Prodotti consigliati
Concentrazione
1 M in toluene
P. ebollizione
111 °C
Densità
0.95 g/mL at 25 °C
Gruppo funzionale
phenyl
Temperatura di conservazione
room temp
Stringa SMILE
[H][C@]12CCCN1B(C)OC2(c3ccccc3)c4ccccc4
InChI
1S/C18H20BNO/c1-19-20-14-8-13-17(20)18(21-19,15-9-4-2-5-10-15)16-11-6-3-7-12-16/h2-7,9-12,17H,8,13-14H2,1H3/t17-/m1/s1
VMKAFJQFKBASMU-QGZVFWFLSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Applicazioni
It may also be used in the preparation of:
- (-)-diospongin B
- (1R)-2-azido-1-(2,2-dimethyl-4H-1,3-benzodioxin-6-yl)ethanol
- (S)-α-deuteriobenzyl alcohol
- (3S,4R,5S)-1-(trimethylsilyl)-4,5-epoxyhex-1-yn-3-ol
Used in a desymmetrizing reduction leading to (S)-4-hydroxycyclohexenone.
Stato fisico
Avvertenze
Danger
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Aquatic Chronic 3 - Asp. Tox. 1 - Eye Dam. 1 - Flam. Liq. 2 - Repr. 2 - Skin Irrit. 2 - STOT RE 2 - STOT SE 3
Organi bersaglio
Central nervous system
Codice della classe di stoccaggio
3 - Flammable liquids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
39.2 °F - closed cup
Punto d’infiammabilità (°C)
4 °C - closed cup
Dispositivi di protezione individuale
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Articoli
Sigma-Aldrich is pleased to offer both enatiomers of 2-methyl-CBS-oxazaborolidine as a dry reagent, in addition to our current offerings as a 1M solution in toluene.
CBS Catalysts
Contenuto correlato
Our company is pleased to offer both enantiomers of 2-methyl-CBS-oxazaborolidine as a dry reagent, in addition to our current offerings as a 1 M solution in toluene.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.