241369
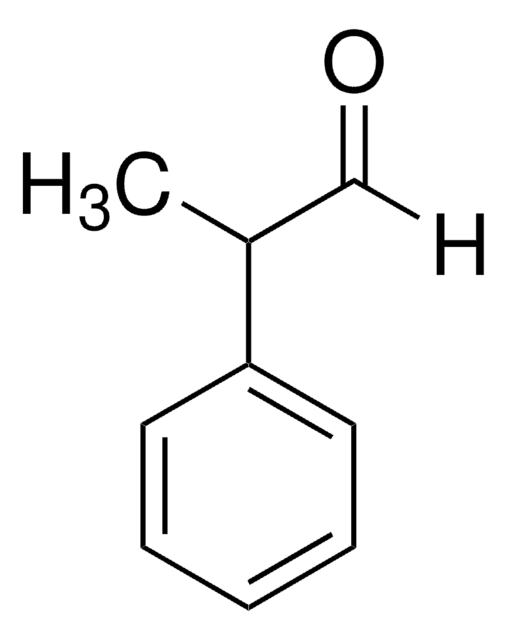
2-Phenylpropionaldehyde
98%
Sinonimo/i:
2-Phenylpropanal, Hydratropaldehyde
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula condensata:
CH3CH(C6H5)CHO
Numero CAS:
Peso molecolare:
134.18
Beilstein:
1905601
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
98%
Stato
liquid
Indice di rifrazione
n20/D 1.517 (lit.)
P. ebollizione
92-94 °C/12 mmHg (lit.)
Densità
1.002 g/mL at 25 °C (lit.)
Gruppo funzionale
aldehyde
phenyl
Temperatura di conservazione
2-8°C
Stringa SMILE
[H]C(=O)C(C)c1ccccc1
InChI
1S/C9H10O/c1-8(7-10)9-5-3-2-4-6-9/h2-8H,1H3
IQVAERDLDAZARL-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Categorie correlate
Applicazioni
2-Phenylpropionaldehyde (hydratropaldehyde) was used as a substrate to study the deformylation activity of reconstituted myoglobin, rMB(1).
Codice della classe di stoccaggio
10 - Combustible liquids
Classe di pericolosità dell'acqua (WGK)
WGK 1
Punto d’infiammabilità (°F)
174.2 °F
Punto d’infiammabilità (°C)
79 °C
Dispositivi di protezione individuale
Eyeshields, Gloves, type ABEK (EN14387) respirator filter
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Takashi Matsuo et al.
Journal of the American Chemical Society, 124(38), 11234-11235 (2002-09-19)
We successfully converted myoglobin, an oxygen-storage hemoprotein, into an oxygen-activating hemoprotein like cytochrome P450s by replacing the native hemin with the artificially created flavohemin. The reconstituted myoglobin, rMb(1), was chacterized by ESI-TOF-mass, UV-vis, and fluorescence spectra. The 1H NMR spectrum
M Popović et al.
Chemical research in toxicology, 17(12), 1568-1576 (2004-12-21)
Felbamate (2-phenyl-1,3-propanediol dicarbamate, FBM) can cause aplastic anemia and hepatotoxicity. The mechanism of FBM-induced toxicities is unknown; however, it has been proposed that 2-phenylpropenal, a reactive metabolite of FBM, is responsible. The pathway leading to this metabolite involves hydrolysis of
C M Dieckhaus et al.
Chemical research in toxicology, 14(5), 511-516 (2001-05-23)
Felbamate has proven to be an effective therapy for treating refractory epilepsy. However, felbamate therapy has been limited due to the associated reports of hepatotoxicity and aplastic anemia. Previous research from our laboratory has proposed 2-phenylpropenal as the reactive metabolite
Shane G Roller et al.
Chemical research in toxicology, 15(6), 815-824 (2002-06-18)
Felbamate is an anti-epileptic drug associated with hepatotoxicity and aplastic anemia. These toxicities are believed to be mediated by the formation of the reactive species 2-phenylpropenal. 4-Hydroxy-5-phenyl-[1,3]oxazinan-2-one is a metabolic precursor for 2-phenylpropenal. 4-Hydroxy-5-phenyl-[1,3]oxazinan-2-one exists in equilibrium with 3-oxo-2-phenylpropyl carbamate
Robert J Parker et al.
Chemical research in toxicology, 18(12), 1842-1848 (2005-12-20)
Evidence has been presented suggesting that a reactive metabolite, 2-phenylpropenal (ATPAL), may be responsible for the toxicities observed during therapy with the antiepileptic drug felbamate (FBM). Formation of ATPAL from its unstable immediate precursor, 3-carbamoyl-2-phenylpropionaldedhyde (CBMA) requires the loss of
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.