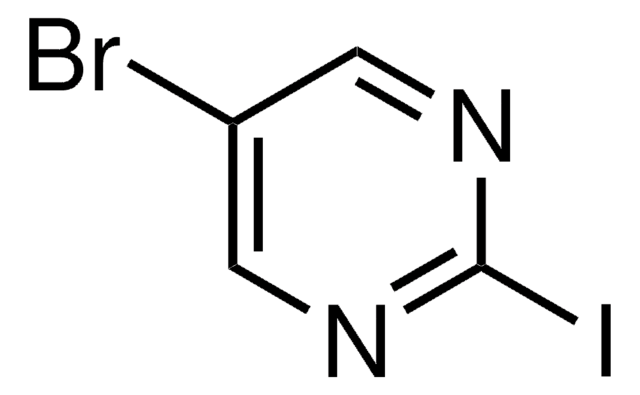
193291
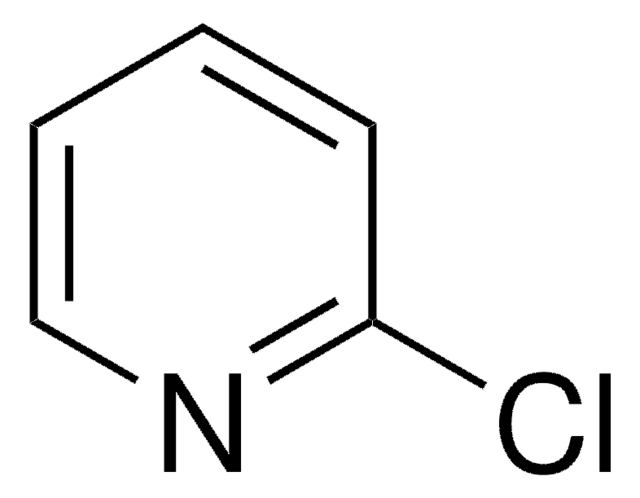
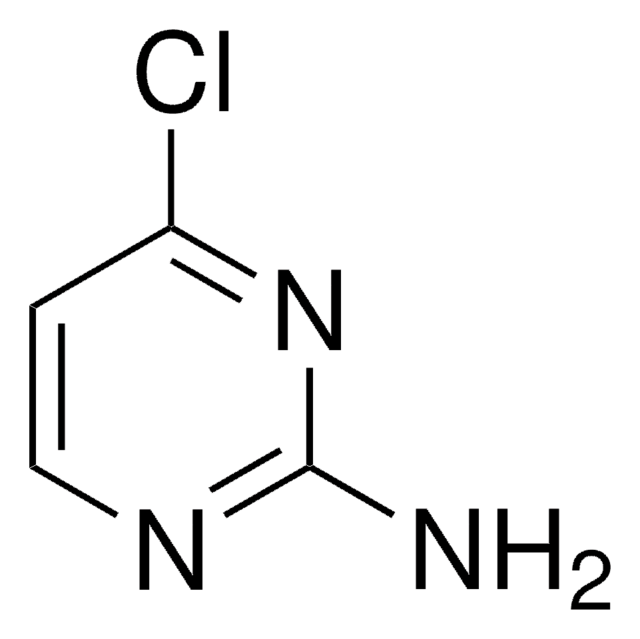
2-Chloropyrimidine
95%
Sinonimo/i:
2-Chloro-1,3-pyrimidine, Pyrimidin-2-yl chloride
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula empirica (notazione di Hill):
C4H3ClN2
Numero CAS:
Peso molecolare:
114.53
Beilstein:
107171
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Livello qualitativo
Saggio
95%
Stato
crystals
P. ebollizione
75-76 °C/10 mmHg (lit.)
Punto di fusione
63-66 °C (lit.)
Gruppo funzionale
chloro
Stringa SMILE
Clc1ncccn1
InChI
1S/C4H3ClN2/c5-4-6-2-1-3-7-4/h1-3H
UNCQVRBWJWWJBF-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Categorie correlate
Descrizione generale
2-Chloropyrimidine undergoes cobalt-catalyzed cross-coupling reaction with aryl halides.
Applicazioni
2-Chloropyrimidine was used in the synthesis of:
- novel bis(2-(pyrimidin-2-yl)ethoxy)alkanes
- 4′-(1,1′-(5-(2-methoxyphenoxy)-[2,2′-bipyrimidine]-4,6-diyl)bis(1H-pyrazol-3,1-diyl)) dianiline fluorescent dye, biosensor for protein assay
- cis- and trans-octahydropyrrolo[2,3]pyridine derivatives
- 2-amino-4-heteroarylpyrimidines
Altre note
Remainder 2-hydroxypyrimidine
Avvertenze
Warning
Indicazioni di pericolo
Consigli di prudenza
Classi di pericolo
Acute Tox. 4 Oral - Eye Irrit. 2
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
208.4 °F - closed cup
Punto d’infiammabilità (°C)
98 °C - closed cup
Dispositivi di protezione individuale
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Vangavaragu Jhansi Rani et al.
Archiv der Pharmazie, 345(8), 663-669 (2012-05-18)
The pyrimidine nucleus is an important component of nucleic acids (DNA and RNA) and vitamins (B(2) and folic acid). It is evident from the literature that pyrimidine derivatives possess a wide spectrum of biological activities such as antioxidant, anticancer, antibacterial
Matthew G Bursavich et al.
Organic letters, 7(19), 4113-4116 (2005-09-09)
[reaction: see text] An expedient synthesis of diverse 2-amino-4-heteroarylpyrimidines via a 2-chloropyrimidine intermediate is described. A series of potentially biologically active analogues have been synthesized in two parallel steps to afford focused arrays.
Igor Goljer et al.
Chirality, 21(7), 681-691 (2008-09-17)
Reaction of (S)- or (R)-3-aminoquinuclidine with 2-chloropyrimidine or 2-bromopyrimidine led to an unexpected formation of both cis- and trans-octahydropyrrolo [2,3]pyridine derivatives. A single-step synthesis of two of the four stereoisomers of these octahydropyrrolo[2,3]pyridine derivatives provides a convenient way of generating
Vikas S Padalkar et al.
Chemistry Central journal, 5, 72-72 (2011-11-10)
Fluorescent dyes with biocompatible functional group and good fluorescence behavior are used as biosensor for monitoring different biological processes as well as detection of protein assay. All reported fluorophore used as sensors are having high selectivity and sensitivity but till
Carina Sollert et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 21(14), 5380-5386 (2015-02-18)
The Ru-catalysed C2-H arylation of indoles and pyrroles by using boronic acids under oxidative conditions is reported. This reaction can be applied to tryptophan derivatives and tolerates a wide range of functional groups on both coupling partners, including bromides and
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.