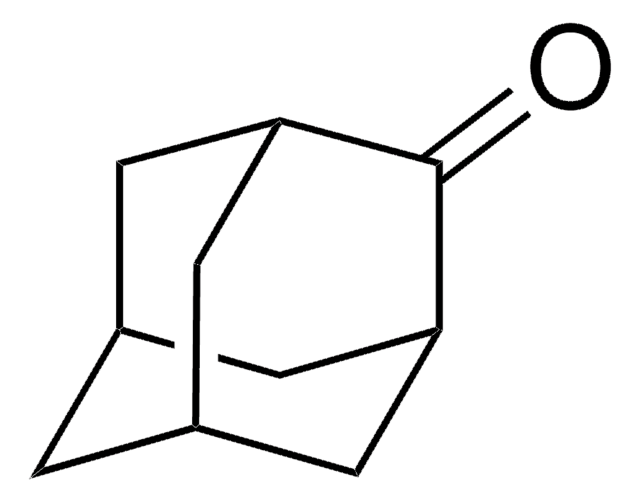
153826
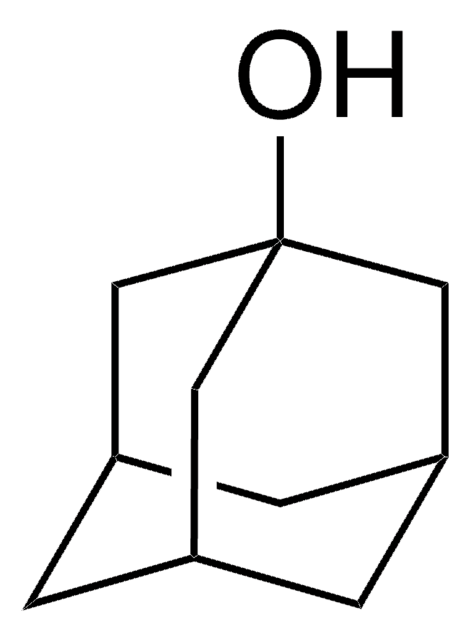
2-Adamantanol
97%
Sinonimo/i:
2-Hydroxyadamantane
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula empirica (notazione di Hill):
C10H16O
Numero CAS:
Peso molecolare:
152.23
Beilstein:
2498536
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Saggio
97%
Stato
solid
Punto di fusione
258-262 °C (lit.)
Gruppo funzionale
hydroxyl
Stringa SMILE
OC1[C@H]2C[C@@H]3C[C@H](C2)C[C@H]1C3
InChI
1S/C10H16O/c11-10-8-2-6-1-7(4-8)5-9(10)3-6/h6-11H,1-5H2/t6-,7+,8-,9+,10?
FOWDOWQYRZXQDP-MGPGSJOLSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Descrizione generale
The low-temperature X-ray structure of 2-adamantanol ester has been studied.
Applicazioni
2-Adamantanol was used to synthesize ester imides.
Codice della classe di stoccaggio
11 - Combustible Solids
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
Not applicable
Punto d’infiammabilità (°C)
Not applicable
Dispositivi di protezione individuale
Eyeshields, Gloves, type N95 (US)
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Andrzej Orzeszko et al.
Farmaco (Societa chimica italiana : 1989), 57(8), 619-624 (2002-10-04)
Some novel ester imides synthesised from trimellitic acid anhydride and 1-adamantanol or 2-adamantanol, were tested as antimicrobial compounds. Unfortunately, these agents showed a modest antibacterial activity (MIC > 6 microg/ml). However, a comparison of these N-substituted adamantylester imides with the
J P Miller et al.
Biochemistry, 33(3), 807-817 (1994-01-25)
Spiro[adamantane-2,2'-diazirine], which produces adamantyl carbene upon photolysis, binds tightly to P450 2B4 (KS = 3.2 microM), giving a normal substrate binding difference spectrum. Irradiation of 2-[3H]adamantane diazirine at 365 nm in the presence of native, ferric P450 2B4 resulted in
Koichi Mitsukura et al.
Applied microbiology and biotechnology, 71(4), 502-504 (2005-09-28)
Hydroxylation of adamantane using whole cells of bacteria, actinomyces, and molds was examined. The structure of the product was determined using gas chromatography (GC), nuclear magnetic resonance (NMR), and mass spectroscopy (MS). Among 470 strains tested, Streptomyces griseoplanus was highly
Marisa Spiniello et al.
Organic & biomolecular chemistry, 1(17), 3094-3101 (2003-10-02)
Results of low-temperature X-ray structural studies for five cis-, and three trans-4-tert-butyl cyclohexanol, and six 2-adamantanol ester and ether derivatives are reported. Plots of C-OR bond distance against pKa(ROH) for derivatives of axial alcohol (5), equatorial alcohol (6) and 2-adamantanol
T Mavromoustakos et al.
Life sciences, 62(20), 1901-1910 (1998-05-26)
Differential Scanning Calorimetry (DSC) has been applied to study the thermal properties of the membrane perturbing antibacterial octyl- and dodecyl-bromide salts of quaternary dimethylamino adamantanol (ADM-8 and ADM-12 correspondingly) incorporated in free or complexed form with beta-cyclodextrin (beta-CD) into dipalmitoylphosphatidylcholine
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.