15369
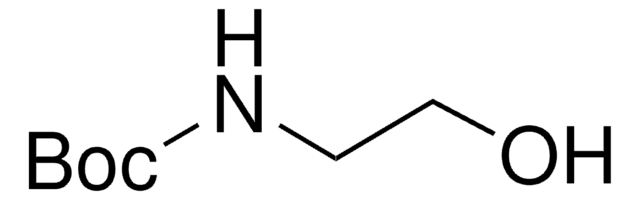
N-Boc-ethylenediamine
≥98.0% (NT)
Sinonimo/i:
N-Boc-1,2-diaminoethane, tert-Butyl N-(2-aminoethyl)carbamate
About This Item
Prodotti consigliati
Livello qualitativo
Saggio
≥98.0% (NT)
Impiego in reazioni chimiche
reagent type: cross-linking reagent
Indice di rifrazione
n20/D 1.458 (lit.)
n20/D 1.458
P. ebollizione
72-80 °C/0.1 mmHg (lit.)
Densità
1.012 g/mL at 20 °C (lit.)
Gruppo funzionale
Boc
amine
Stringa SMILE
NCCNC(OC(C)(C)C)=O
InChI
1S/C7H16N2O2/c1-7(2,3)11-6(10)9-5-4-8/h4-5,8H2,1-3H3,(H,9,10)
AOCSUUGBCMTKJH-UHFFFAOYSA-N
Cerchi prodotti simili? Visita Guida al confronto tra prodotti
Applicazioni
- Facile synthesis of new N-(aminocycloalkylene) amino acid compounds: Describes the use of N-Boc-ethylenediamine in the preparation of benzyl 2-{[2-(Boc-amino)ethyl]amino}acetate (G Matulevičiūtė et al., 2023).
- Synthesis of a new tripod BODIPY dye: Reports on the chemical synthesis involving N-Boc-ethylenediamine for creating a BODIPY dye (H Wang et al., 2022).
- Single‐Entity Electrocatalysis: Discusses the application of N-Boc-ethylenediamine in modifying Co3O4 nanoparticles used in electrocatalysis (T Quast et al., 2021).
Altre note
Avvertenze
Danger
Indicazioni di pericolo
Classi di pericolo
Skin Corr. 1B
Codice della classe di stoccaggio
8A - Combustible corrosive hazardous materials
Classe di pericolosità dell'acqua (WGK)
WGK 3
Punto d’infiammabilità (°F)
235.4 °F - closed cup
Punto d’infiammabilità (°C)
113 °C - closed cup
Dispositivi di protezione individuale
Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter
Scegli una delle versioni più recenti:
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.