PHR1526
Dexaméthasone
Pharmaceutical Secondary Standard; Certified Reference Material
Synonyme(s) :
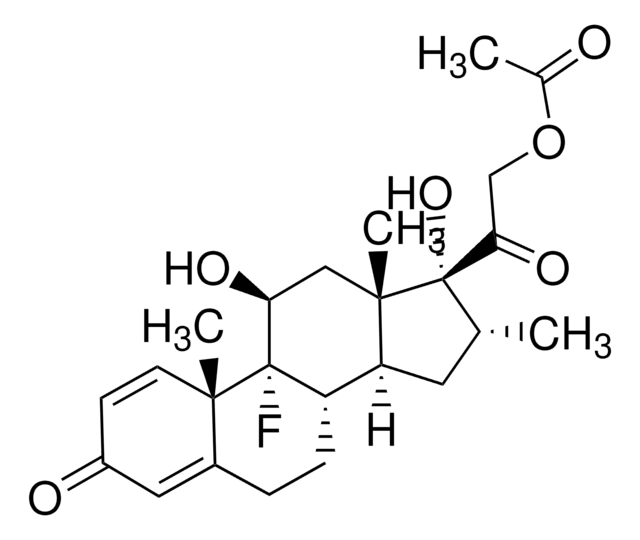
(11β,16α)-9-Fluoro-11,17,21-trihydroxy-16-méthylpregna-1,4-diène-3,20-dione, 9α-Fluoro-16α-méthyl-11β,17α,21-trihydroxy-1,4-pregnadiène-3,20-dione, 9α-Fluoro-16α-méthylprednisolone, Prednisolone F
About This Item
Produits recommandés
Source biologique
synthetic
Niveau de qualité
Qualité
certified reference material
pharmaceutical secondary standard
Agence
BP
EP
USP
traceable to BP 578
traceable to Ph. Eur. D0700000
traceable to USP 1176007
Pression de vapeur
<0.0000001 kPa ( 25 °C)
Famille d'API
dexamethasone
CofA (certificat d'analyse)
current certificate can be downloaded
Conditionnement
pkg of 1 g
Conditions de stockage
protect from light
Technique(s)
HPLC: suitable
gas chromatography (GC): suitable
Couleur
white
Pf
262-264 °C (lit.)
Solubilité
DMSO: soluble
acetone: sparingly soluble
chloroform: slightly soluble
dioxane: soluble
ethanol: sparingly soluble
ether: very slightly soluble
methanol: sparingly soluble
water: practically insoluble
Application(s)
pharmaceutical (small molecule)
Format
neat
Conditions d'expédition
ambient
Température de stockage
2-30°C
Chaîne SMILES
C[C@@H]1C[C@H]2[C@@H]3CCC4=CC(=O)C=C[C@]4(C)[C@@]3(F)[C@@H](O)C[C@]2(C)[C@@]1(O)C(=O)CO
InChI
1S/C22H29FO5/c1-12-8-16-15-5-4-13-9-14(25)6-7-19(13,2)21(15,23)17(26)10-20(16,3)22(12,28)18(27)11-24/h6-7,9,12,15-17,24,26,28H,4-5,8,10-11H2,1-3H3/t12-,15+,16+,17+,19+,20+,21+,22+/m1/s1
Clé InChI
UREBDLICKHMUKA-CXSFZGCWSA-N
Informations sur le gène
human ... NR3C1(2908)
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Description générale
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
Application
Actions biochimiques/physiologiques
Remarque sur l'analyse
Autres remarques
Note de bas de page
Produits recommandés
Produit(s) apparenté(s)
Mention d'avertissement
Danger
Mentions de danger
Conseils de prudence
Classification des risques
Repr. 1B
Code de la classe de stockage
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique