176141
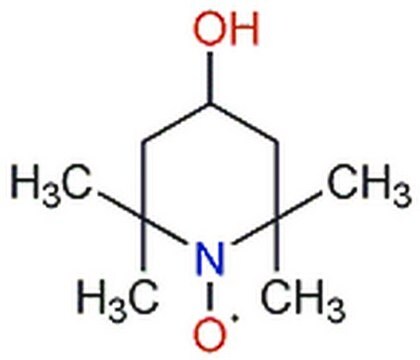
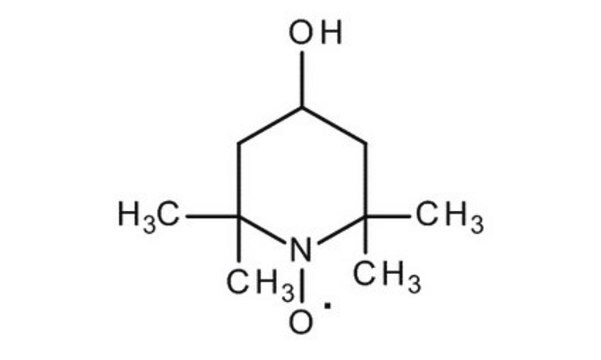
4-Hydroxy-TEMPO
95%
Synonyme(s) :
4-Hydroxy-2,2,6,6-tetramethylpiperidine 1-oxyl, TEMPOL
About This Item
Produits recommandés
Niveau de qualité
Essai
95%
Forme
solid
Composition
Carbon content, 60.6-65.0%
Nitrogen content, 7.8-8.4%
Pf
69-71 °C (lit.)
Groupe fonctionnel
hydroxyl
Température de stockage
2-8°C
Chaîne SMILES
CC1(C)CC(O)CC(C)(C)N1[O]
InChI
1S/C9H18NO2/c1-8(2)5-7(11)6-9(3,4)10(8)12/h7,11H,5-6H2,1-4H3
Clé InChI
UZFMOKQJFYMBGY-UHFFFAOYSA-N
Vous recherchez des produits similaires ? Visite Guide de comparaison des produits
Description générale
Application
Mention d'avertissement
Danger
Mentions de danger
Conseils de prudence
Classification des risques
Acute Tox. 4 Oral - Eye Dam. 1 - STOT RE 2 Oral
Organes cibles
Liver,spleen
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 1
Point d'éclair (°F)
294.8 °F - DIN 51758
Point d'éclair (°C)
146 °C - DIN 51758
Équipement de protection individuelle
dust mask type N95 (US), Eyeshields, Gloves
Faites votre choix parmi les versions les plus récentes :
Certificats d'analyse (COA)
Vous ne trouvez pas la bonne version ?
Si vous avez besoin d'une version particulière, vous pouvez rechercher un certificat spécifique par le numéro de lot.
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Articles
TEMPO (2,2,6,6-Tetramethylpiperidinyloxy or 2,2,6,6-Tetramethylpiperidine 1-oxyl) and its derivatives are stable nitroxy radicals used as catalysts in organic oxidation reactions. TEMPO was discovered by Lebedev and Kazarnovskii in 1960. The stable free radical nature of TEMPO is due to the presence of bulky substituent groups, which hinder the reaction of the free radical with other molecules.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique