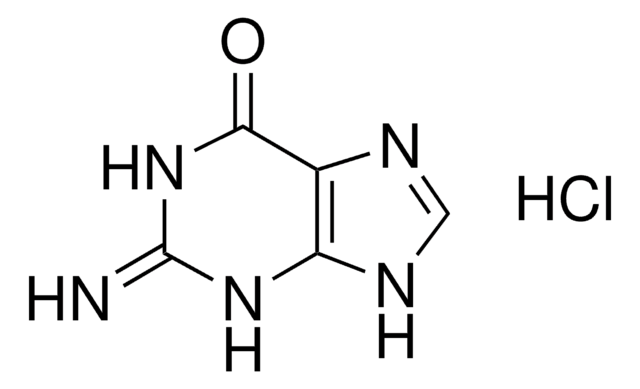
G3272
Guanidine hydrochloride
for molecular biology, ≥99%
Synonym(s):
Aminoformamidine hydrochloride, Aminomethanamidine hydrochloride, Guanidinium chloride
About This Item
Recommended Products
biological source
synthetic (organic)
Quality Level
grade
for molecular biology
Assay
≥99%
form
crystalline powder
storage condition
(Tightly closed. Dry. )
technique(s)
RNA extraction: suitable
impurities
≤0.3% water (Karl Fischer)
color
white
pH
(25 °C, 4.6 - 6/573 g/L)
mp
180-185 °C (lit.)
solubility
H2O: 6 M, clear, colorless
density
1.3 g/cm3 (lit.)
cation traces
Pb: ≤5 ppm
UV absorption
λ: 260 nm Amax: ≤0.10
λ: 290 nm Amax: ≤0.05
foreign activity
DNase, RNase, none detected
SMILES string
Cl[H].NC(N)=N
InChI
1S/CH5N3.ClH/c2-1(3)4;/h(H5,2,3,4);1H
InChI key
PJJJBBJSCAKJQF-UHFFFAOYSA-N
Gene Information
human ... KCNA1(3736) , KCNA10(3744) , KCNA2(3737) , KCNA3(3738) , KCNA4(3739) , KCNA5(3741) , KCNA6(3742) , KCNA7(3743) , KCNB1(3745) , KCNB2(9312) , KCNC1(3746) , KCNC2(3747) , KCNC3(3748) , KCNC4(3749) , KCND1(3750) , KCND2(3751) , KCND3(3752) , KCNF1(3754) , KCNG1(3755) , KCNG2(26251) , KCNG3(170850) , KCNG4(93107) , KCNH1(3756) , KCNH2(3757) , KCNH3(23416) , KCNH4(23415) , KCNH5(27133) , KCNH6(81033) , KCNH7(90134) , KCNH8(131096) , KCNQ1(3784) , KCNQ2(3785) , KCNQ3(3786) , KCNQ4(9132) , KCNQ5(56479) , KCNS1(3787) , KCNS2(3788) , KCNS3(3790) , KCNV1(27012) , KCNV2(169522)
Looking for similar products? Visit Product Comparison Guide
General description
At lower concentrations, guanidine hydrochloride exhibits a unique effect, promoting the refolding of denatured proteins and restoring enzymatic activity. This property is advantageous for protein renaturation studies. In RNA extraction, guanidine hydrochloride acts as a robust denaturant, disrupting cell structures and inactivating RNA enzymes, ensuring the integrity of extracted RNA. In summary, guanidine hydrochloride is a versatile compound with applications in protein purification, nucleic acid isolation, and protein refolding studies, showcasing its multifaceted utility in molecular biology and biochemical research.
Application
- as a component of the extraction buffer for the extraction of proteoglycans
- in extraction during protein fractionation of ATDC5 cell lines
- as a chemical additive to study its effective absorbance spectra in structural analysis
- to incubate the device chip to release prostate-specific antigen and for regeneration of aptamer
- as a component of the 2,4-dinitrophenylhydrazine (DNPH) solution for detecting protein carbonyls by protein carbonylation study
- in RNA isolation to dissociate nucleoproteins and inhibit RNase
Biochem/physiol Actions
Features and Benefits
- Highly versatile surfactant for your cell biology and biochemical research
- Suitable for sensitive molecular biology applications
- Free from Exonuclease, Dnase and Rnase
- Tested to confirm low levels of heavy metal contamination, ensuring suitability for various applications
Caution
Preparation Note
The maximum solubility of guanidine hydrochloride in water at room temperature is approximately 6M.
Other Notes
comparable product
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service