SML0011
VAHA
≥98% (HPLC)
Sinônimo(s):
VAHA, Valproyl hydroxamic acid, VPA-HA
About This Item
Produtos recomendados
Nível de qualidade
Ensaio
≥98% (HPLC)
forma
powder
solubilidade
DMSO: ≥25 mg/mL
temperatura de armazenamento
2-8°C
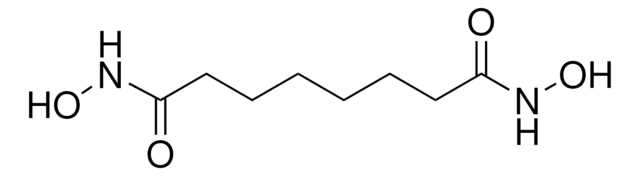
cadeia de caracteres SMILES
CCCC(CCC)C(=O)NO
InChI
1S/C8H17NO2/c1-3-5-7(6-4-2)8(10)9-11/h7,11H,3-6H2,1-2H3,(H,9,10)
chave InChI
ROJGIRXXBBBMPL-UHFFFAOYSA-N
Aplicação
Ações bioquímicas/fisiológicas
Características e benefícios
Palavra indicadora
Warning
Frases de perigo
Declarações de precaução
Classificações de perigo
Acute Tox. 4 Oral
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Certificados de análise (COA)
Busque Certificados de análise (COA) digitando o Número do Lote do produto. Os números de lote e remessa podem ser encontrados no rótulo de um produto após a palavra “Lot” ou “Batch”.
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Artigos
Epigenetic modifications are thought to occur through two key interconnected processes—DNA methylation and the covalent modification of histones.
Conteúdo relacionado
We offer a variety of small molecule research tools, such as transcription factor modulators, inhibitors of chromatin modifying enzymes, and agonists/antagonists for target identification and validation in gene regulation research; a selection of these research tools is shown below.
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica