D5782
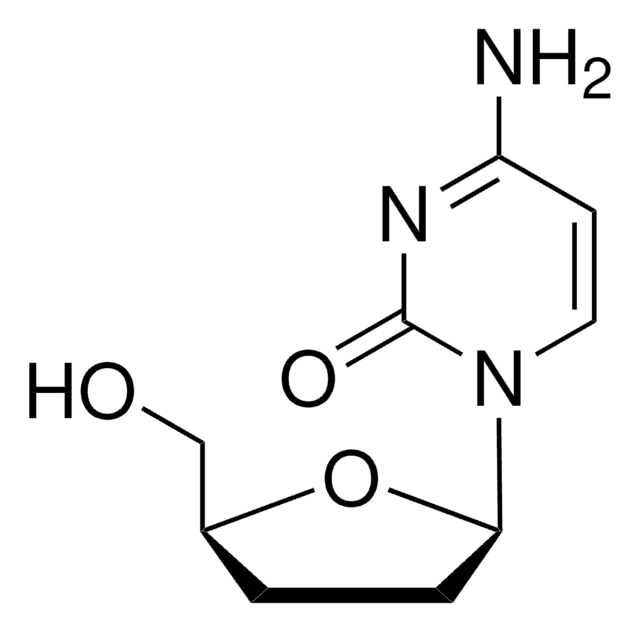
2′,3′-Dideoxycytidine
≥98% (HPLC)
Sinônimo(s):
ddC
About This Item
Produtos recomendados
fonte biológica
synthetic (organic)
Ensaio
≥98% (HPLC)
Formulário
powder
cor
colorless
pf
217-218 °C (lit.)
solubilidade
water: 50 mg/mL, clear, colorless to faintly yellow
temperatura de armazenamento
−20°C
cadeia de caracteres SMILES
NC1=NC(=O)N(C=C1)[C@H]2CC[C@@H](CO)O2
InChI
1S/C9H13N3O3/c10-7-3-4-12(9(14)11-7)8-2-1-6(5-13)15-8/h3-4,6,8,13H,1-2,5H2,(H2,10,11,14)/t6-,8+/m0/s1
chave InChI
WREGKURFCTUGRC-POYBYMJQSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Aplicação
- as a DNA chain-terminating nucleotide for DNA sequencing methods based on the Sanger chain-termination method
- as a nucleoside reverse transcriptase inhibitor (NRTI) to study its effects on the development of mechanical allodynia in aging mice
- as a mitochondrial DNA (mtDNA) replication inhibitor to inhibit the activation of cGAS-STING pathway and study its effects on signaling protein-stimulator of interferon genes (STING), cyclic GMP-AMP synthase (cGAS), and phospho-interferon regulator factor 3 (p-IRF3) expression in mouse hippocampal and microglial cells
- as an NRTI inhibitor to study its effects on the drug induced-mitochondrial toxicity in Caenorhabditis elegans
Ações bioquímicas/fisiológicas
Palavra indicadora
Warning
Frases de perigo
Declarações de precaução
Classificações de perigo
Carc. 2
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
Eyeshields, Gloves, type P3 (EN 143) respirator cartridges
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica