D87589
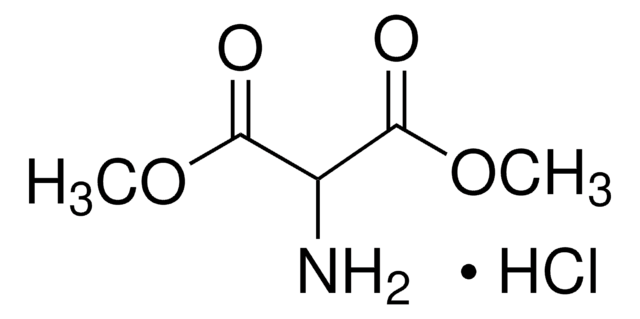
Diethyl aminomalonate hydrochloride
98%
Sinônimo(s):
Aminomalonic acid diethyl ester hydrochloride
Faça loginpara ver os preços organizacionais e de contrato
About This Item
Fórmula linear:
NH2CH2(COOC2H5)2 · HCl
Número CAS:
Peso molecular:
211.64
Beilstein:
3568037
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de substância PubChem:
NACRES:
NA.22
Produtos recomendados
Nível de qualidade
Ensaio
98%
Formulário
crystals
pf
165-170 °C (dec.) (lit.)
cadeia de caracteres SMILES
Cl.CCOC(=O)C(N)C(=O)OCC
InChI
1S/C7H13NO4.ClH/c1-3-11-6(9)5(8)7(10)12-4-2;/h5H,3-4,8H2,1-2H3;1H
chave InChI
GLFVNTDRBTZJIY-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
Not applicable
Ponto de fulgor (°C)
Not applicable
Equipamento de proteção individual
dust mask type N95 (US), Eyeshields, Gloves
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Os clientes também visualizaram
C M Metzler et al.
Biochemistry, 27(13), 4923-4933 (1988-06-28)
To establish the state of protonation of quinonoid species formed nonenzymically from pyridoxal phosphate (PLP) and diethyl aminomalonate, we have studied absorption spectra of the rapidly established steady-state mixture of species. We have evaluated the formation constant and the spectrum
Seiichi Ohta et al.
Molecular pharmaceutics, 14(9), 3105-3113 (2017-08-15)
Intraperitoneal administration of chemotherapeutics is expected for the treatment of peritoneally disseminated gastric cancer because of poor migration of the drugs from the systemic circulation to the peritoneal cavity. In this study, for intraperitoneal delivery of cisplatin (CDDP), we developed
Reaction control in the organocatalytic asymmetric one-pot, three-component reaction of aldehydes, diethyl alpha-aminomalonate and nitroalkenes: toward diversity-oriented synthesis.
Yan-Kai Liu et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 14(32), 9873-9877 (2008-10-04)
F Hughes et al.
Organic letters, 3(18), 2911-2914 (2001-09-01)
[reaction: see text]. Nitrogen-containing tethered diacids, easily prepared by reductive alkylation of diethyl aminomalonate or ethyl cyanoglycinate, undergo double Michael reactions with 3-butyn-2-one to give highly functionalized and substituted piperidines (pipecolic acid derivatives) with surprisingly high stereoselectivity. The heterocyclic double
Charles M Blazey et al.
The Journal of organic chemistry, 67(1), 298-300 (2002-01-05)
The azomethine ylide derived from the condensation of diethyl aminomalonate with paraformaldehyde undergoes 1,3-dipolar cycloadditions with acrylate and propiolate derivatives. Contrary to a previous report, these reactions yield mixtures of regioisomers generally favoring the 2,2,3-trisubstituted product. However, the relative quantity
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica