About This Item
Fórmula empírica (Notação de Hill):
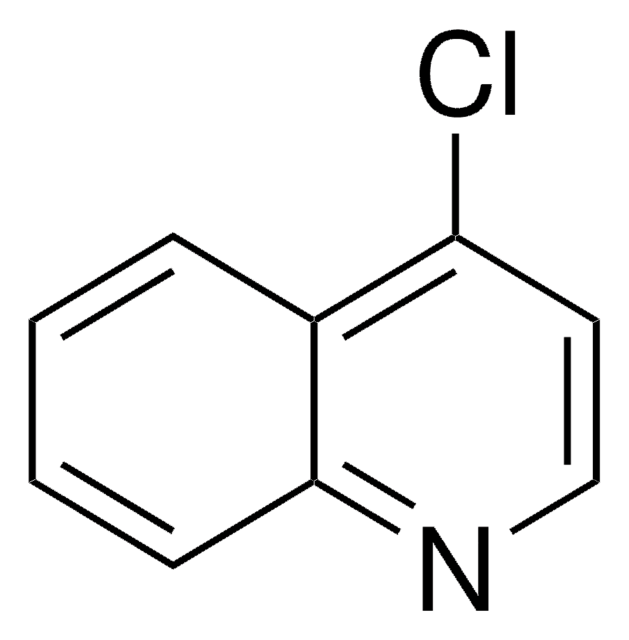
C9H6ClN
Número CAS:
Peso molecular:
163.60
Beilstein:
112561
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de substância PubChem:
NACRES:
NA.22
Produtos recomendados
Ensaio
99%
Formulário
crystals
p.e.
266-267 °C (lit.)
pf
34-37 °C (lit.)
densidade
1.23 g/mL at 25 °C (lit.)
cadeia de caracteres SMILES
Clc1ccc2ccccc2n1
InChI
1S/C9H6ClN/c10-9-6-5-7-3-1-2-4-8(7)11-9/h1-6H
chave InChI
OFUFXTHGZWIDDB-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Palavra indicadora
Warning
Frases de perigo
Declarações de precaução
Classificações de perigo
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Órgãos-alvo
Respiratory system
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
230.0 °F - closed cup
Ponto de fulgor (°C)
110 °C - closed cup
Equipamento de proteção individual
dust mask type N95 (US), Eyeshields, Gloves
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
K Shiva Kumar et al.
Bioorganic & medicinal chemistry, 20(7), 2199-2207 (2012-03-06)
A number of 2-(1H-indol-3-yl)quinoline-3-carbonitrile derivatives were synthesized via AlCl(3)-mediated C-C bond forming reaction between 2-chloroquinoline-3-carbonitrile and various indoles. The methodology does not require any N-protection of the indoles employed and provided the corresponding products in good yields. The molecular structure
Santosh Kumar et al.
International journal of biological macromolecules, 49(3), 356-361 (2011-06-07)
This paper describes an elegant cross-linking technique for the preparation of chitosan-chloroquinoline derivative by using a greener technique. Chitosan solution in aqueous acetic acid was treated with 2-chloroquinoline-3-carbaldehyde solution to form hydrogel; the resulting hydrogel was subjected to solvent exchange.
S Fetzner et al.
FEMS microbiology letters, 112(2), 151-157 (1993-09-01)
Resting cells of Pseudomonas putida strain 86 were grown on quinoline transformed 2-chloroquinoline to 2-chloro-cis-7,8-dihydro-7,8-dihydroxyquinoline which was not converted further. 7,8-Dioxygenating activity was present when the enzymes of quinoline catabolism were induced. Quinoline-grown cells of strain 86 treated simultaneously with
Krzysztof Marciniec et al.
Journal of chromatographic science, 55(9), 934-939 (2017-06-28)
The lipophilicity of a series of anticancer propargylquinoline derivatives is investigated using both chromatographic and computational methods. The parameters of the tested compounds' relative lipophilicity (logkw) are determined experimentally by the high-performance liquid chromatographic method (RP-HPLC, Accucore C18 column), using
D R Boyd et al.
Organic & biomolecular chemistry, 8(5), 1081-1090 (2010-02-19)
A series of enantiopure 2,2'-bipyridines have been synthesised from the corresponding cis-dihydrodiol metabolites of 2-chloroquinolines. Several of the resulting hydroxylated 2,2'-bipyridines were found to be useful chiral ligands for the asymmetric aminolysis of meso-epoxides leading to the formation of enantioenriched
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica