123323
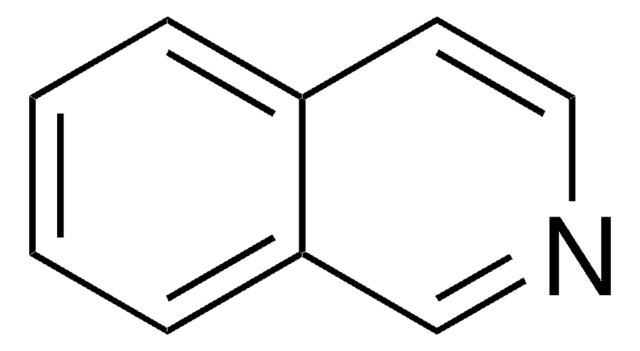
Quinazoline
99%
Sinônimo(s):
1,3-Benzodiazine, Benzopyrimidine
Faça loginpara ver os preços organizacionais e de contrato
About This Item
Fórmula empírica (Notação de Hill):
C8H6N2
Número CAS:
Peso molecular:
130.15
Beilstein:
109370
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de substância PubChem:
NACRES:
NA.22
Produtos recomendados
Nível de qualidade
Ensaio
99%
Formulário
solid
p.e.
243 °C (lit.)
pf
46-48 °C (lit.)
solubilidade
H2O: freely soluble
organic solvents: soluble
cadeia de caracteres SMILES
c1ccc2ncncc2c1
InChI
1S/C8H6N2/c1-2-4-8-7(3-1)5-9-6-10-8/h1-6H
chave InChI
JWVCLYRUEFBMGU-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Categorias relacionadas
Descrição geral
Quinazolines has applications in medicinal chemistry due to their antibacterial, antifungal, anticonvulsant, anti-inflammatory and antitumor activities. It is the basic structural unit of pharmaceuticals and plays an important role in modern synthesis of antitumor drugs.
Aplicação
Quinazoline was used to study the electrochemical behaviour of quinazoline using modern polarographic and voltammetric methods.
Ações bioquímicas/fisiológicas
Genotoxicity of quinazoline was established by bacterial SOS Chromotest (Escherichia Coli).
Código de classe de armazenamento
11 - Combustible Solids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
222.8 °F - closed cup
Ponto de fulgor (°C)
106 °C - closed cup
Equipamento de proteção individual
Eyeshields, Gloves, type N95 (US)
Escolha uma das versões mais recentes:
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Zhijiang Fan et al.
Molecular diversity, 22(3), 657-667 (2018-03-27)
A series of structurally novel 3-thioether-1-(quinazolin-4-yl)-1H-1,2,4-triazol-5-amine derivatives (7a-7r) were designed and synthesized based on a pharmacophore hybrid approach, and screened for their antibacterial and antifungal activities in vitro. All the target compounds were fully characterized through [Formula: see text]H NMR
Elham Bagheri et al.
Current pharmaceutical design, 24(13), 1395-1404 (2018-02-01)
Quinazoline is an aromatic bicyclic compound exhibiting several pharmaceutical and biological activities. This study was conducted to investigate the potential wound healing properties of Synthetic Quinazoline Compound (SQC) on experimental rats. The toxicity of SQC was determined by MTT cell
Siva Jyothi Buggana et al.
Computational biology and chemistry, 79, 110-118 (2019-02-21)
Recent studies reported the involvement of JAK2/STAT3 pathway in various solid tumours including breast, ovarian, prostate and lung cancers. Clinical literature also reported the lowered burden in breast and ovarian cancers by targeting JAK2 pathway. In this study, a series
Polarographic and voltammetric determination of quinazoline-the structural unit of anticancer drugs.
Hladikova J, et al.
Sensing in Electroanalysis, 3, 165-175 (2008)
Reddy Amala et al.
BioImpacts : BI, 11(1), 15-22 (2021-01-21)
Introduction: Inflammation is the primary response caused due to harmful stimuli which are followed by the increased draining of plasma and immune cells from the body into the site of the injured tissue. A signaling cascade of growth factors and
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica