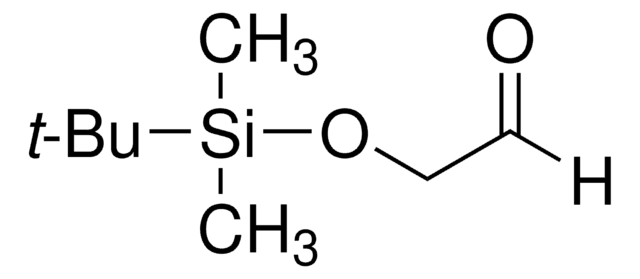
497401
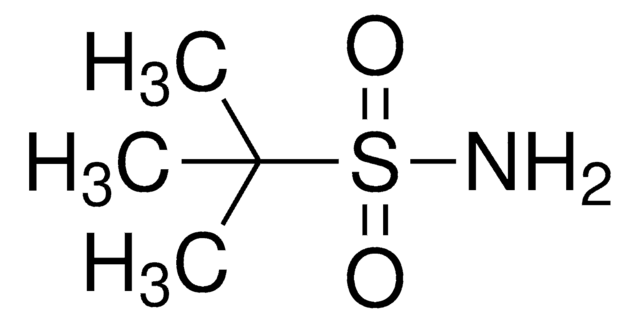
(R)-(+)-2-Methyl-2-propanesulfinamide
98%
Synonyme(s) :
(R)-2-methyl-2-propanesulfinamide, (R)-2-methylpropane-2-sulfinamide, (R)-tert-butanesulfinamide, (R)-tert-butylsulfinamide
About This Item
Produits recommandés
Pureté
98%
Activité optique
[α]20/D +4°, c = 1.0242 in chloroform stab. with amylenes
Pf
103-107 °C (lit.)
Température de stockage
2-8°C
Chaîne SMILES
CC(C)(C)S(N)=O
InChI
1S/C4H11NOS/c1-4(2,3)7(5)6/h5H2,1-3H3/t7-/m1/s1
Clé InChI
CESUXLKAADQNTB-SSDOTTSWSA-N
Catégories apparentées
Description générale
Application
Code de la classe de stockage
11 - Combustible Solids
Classe de danger pour l'eau (WGK)
WGK 3
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
Eyeshields, Gloves, type N95 (US)
Faites votre choix parmi les versions les plus récentes :
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Les clients ont également consulté
Articles
Ellman's sulfinamide is available in both enantiomeric and racemic forms for your research. This versatile and useful auxiliary has found extensive use both in academics and industry.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique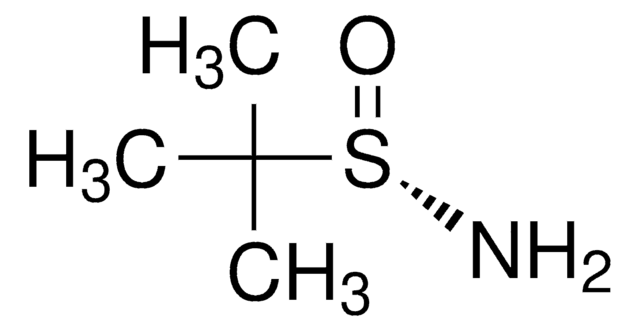



![(R)-N-[(1R,2R)-2-(3-(3,5-Bis(trifluoromethyl)phenyl)ureido)cyclohexyl]-tert-butyl-sulfinamide 96%](/deepweb/assets/sigmaaldrich/product/structures/389/070/18847164-c6a7-4b4e-abcb-2dbc22493a2d/640/18847164-c6a7-4b4e-abcb-2dbc22493a2d.png)