Alle Fotos(1)
Wichtige Dokumente
I0404
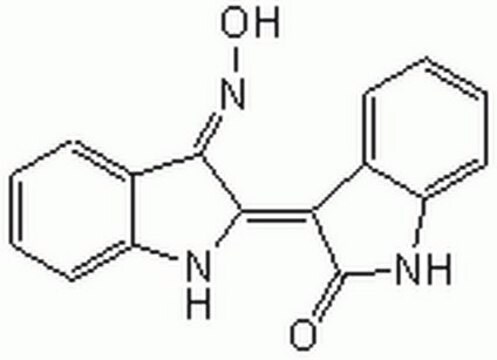
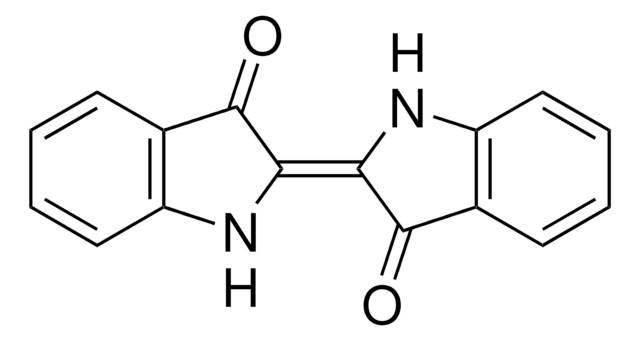
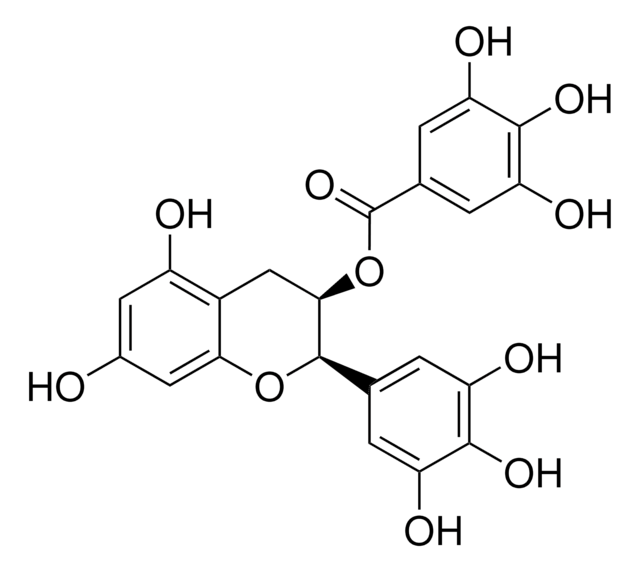
Indirubin-3′-oxime
≥98% (HPLC), solid
Synonym(e):
Indirubin-3′-monoxime
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(1)
About This Item
Empirische Formel (Hill-System):
C16H11N3O2
CAS-Nummer:
Molekulargewicht:
277.28
MDL-Nummer:
UNSPSC-Code:
12352200
PubChem Substanz-ID:
NACRES:
NA.77
Empfohlene Produkte
Qualitätsniveau
Assay
≥98% (HPLC)
Form
solid
Mol-Gew.
277.28
Löslichkeit
DMSO: >10 mg/mL
H2O: insoluble
Lagertemp.
room temp
SMILES String
O\N=C1\C(Nc2ccccc12)=C3\C(=O)Nc4ccccc34
InChI
1S/C16H11N3O2/c20-16-13(9-5-1-3-7-11(9)18-16)15-14(19-21)10-6-2-4-8-12(10)17-15/h1-8,17,21H,(H,18,20)/b15-13-,19-14+
InChIKey
HBDSHCUSXQATPO-BGBJRWHRSA-N
Anwendung
Indirubin-3′-oxime has been used in the inhibition of glycogen synthase kinase 3 in human monocytic cell line, THP-1.
Biochem./physiol. Wirkung
Indirubin-3′-oxime is a cyclin-dependent kinase inhibitor which functions by competing with ATP for binding to the catalytic subunit; exhibits antiproliferative activity leading to G2/M arrest in many cell lines and G1/S arrest in Jurkat cells.
Indirubin-3′-oxime mediates apoptosis in Jurkat T cells and has anti-tumor functionality. Indirubin-3′-oxime inhibits Y box binding protein 1 (YB1) translocation, contributing to anticancer functionality. Indirubin-3′-oxime decreases expression of estrogen-related receptor γ (ERRγ) and peroxisome proliferator-activated receptor-γ co-activator-1α (PGC1α) in human neuroblastoma, leading to cell cycle arrest and mitochondrial dysfunction.
Leistungsmerkmale und Vorteile
This compound is featured on the CDKs and GSK-3 pages of the Handbook of Receptor Classification and Signal Transduction. To browse other handbook pages, click here.
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
Eyeshields, Gloves, type N95 (US)
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Indirubin-3'-oxime induces mitochondrial dysfunction and triggers growth inhibition and cell cycle arrest in human neuroblastoma cells.
Liao XM and Leung KN
Oncology Reports, 29(1), 371-379 (2013)
Dynamic modulation of innate immune response by varying dosages of LPS in human monocytic cells.
Morris M, et al.
The Journal of Biological Chemistry, 289(31), 21584-21590 (2014)
Mechanism responsible for inhibitory effect of indirubin 3?-oxime on anticancer agent-induced YB-1 nuclear translocation in HepG2 human hepatocellular carcinoma cells.
Tanaka T, et al.
Experimental Cell Research, 370(2), 454-460 (2018)
Semi-synthesis of indirubin-3′-oxime from Strobilanthes cusia leaves, its acute and sub-chronic toxicity, in vitro and in vivo antitumor activity in Lewis lung carcinoma bearing mice.
Cuong NM, et al.
Journal of Pharmacognosy and Phytochemistry, 5(4), 205-205 (2016)
Ming-Yang Lee et al.
European journal of pharmacology, 839, 57-65 (2018-09-30)
Cholangiocarcinoma (CCA) is one of the most serious of all cancers and a major public health problem. CCA is an extremely invasive cancer, and the survival rate for CCA patients is only 24 months after diagnosis. Although surgery and chemotherapy
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.