275778
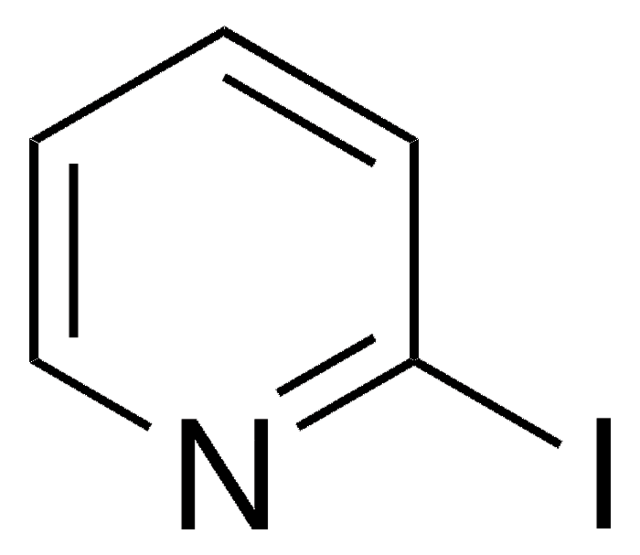
Imidazo[1,2-a]pyridin
99%
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(1)
About This Item
Empirische Formel (Hill-System):
C7H6N2
CAS-Nummer:
Molekulargewicht:
118.14
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Assay
99%
Form
liquid
Brechungsindex
n20/D 1.626 (lit.)
bp
103 °C/1 mmHg (lit.)
Dichte
1.165 g/mL at 25 °C (lit.)
SMILES String
c1ccn2ccnc2c1
InChI
1S/C7H6N2/c1-2-5-9-6-4-8-7(9)3-1/h1-6H
InChIKey
UTCSSFWDNNEEBH-UHFFFAOYSA-N
Verwandte Kategorien
Allgemeine Beschreibung
In vivo anti-trypanosomal activity of imidazo[1,2-a]pyridiness in the STIB900 mouse model has been investigated.
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Zielorgane
Respiratory system
Lagerklassenschlüssel
10 - Combustible liquids
WGK
WGK 3
Flammpunkt (°F)
235.4 °F - closed cup
Flammpunkt (°C)
113 °C - closed cup
Persönliche Schutzausrüstung
Eyeshields, Gloves, type ABEK (EN14387) respirator filter
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Suren Husinec et al.
Organic letters, 13(9), 2286-2289 (2011-03-31)
A base promoted cyclization of the protected N-propargylaminopyridines was shown to be an efficient method for the preparation of imidazo[1,2-a]pyridine derivatives. The reactions were carried out with a small excess of base, at room temperature or slightly above producing the
Richard Ducray et al.
Bioorganic & medicinal chemistry letters, 21(16), 4698-4701 (2011-07-22)
We disclose a novel series of insulin-like growth factor-1 receptor kinase inhibitors based on the 3-(pyrimidin-4-yl)-imidazo[1,2-a]pyridine scaffold. The influence on the inhibitory activity of substitution on the imidazopyridine and at the C5 position of the pyrimidine is discussed. In the
Ebrahim Kianmehr et al.
Journal of combinatorial chemistry, 12(1), 41-44 (2009-11-13)
A one-pot, three-component reaction between pyridine, phenacyl bromide, and thiocyanate is described. The reaction afforded the corresponding special type of fully substituted imidazo[1,2-a]pyridine derivatives in good yields without using any catalyst or activation.
Richard Ducray et al.
Bioorganic & medicinal chemistry letters, 21(16), 4702-4704 (2011-07-19)
Following the discovery of imidazopyridine 1 as a potent IGF-1R tyrosine kinase inhibitor, the aniline part has been modified with the aim to optimize the properties of this series. The structure-activity relationships against IGF-1R kinase activity as well as inhibition
Hongpeng Sun et al.
The Journal of organic chemistry, 77(23), 10745-10751 (2012-11-22)
An efficient tandem route to the synthesis of 3H-1,2a(1),3-triazaacenaphthylene derivatives of the cyclazine family has been developed. Target compounds were obtained in moderate to good yields by a Yb(OTf)(3)/Ag(2)CO(3)-catalyzed, three-component domino reaction. This in turn will set the stage for
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.![2-phenylimidazo[1,2-a]pyridine AldrichCPR](/deepweb/assets/sigmaaldrich/product/structures/281/247/6c2550a0-2f0c-4866-83d8-3c1fb039e165/640/6c2550a0-2f0c-4866-83d8-3c1fb039e165.png)

![Imidazo[1,2-a]pyrazin 97%](/deepweb/assets/sigmaaldrich/product/structures/370/804/1712d71f-52fb-4758-9a22-85b6c96cd4e8/640/1712d71f-52fb-4758-9a22-85b6c96cd4e8.png)
![Imidazo[1,2-a]pyrimidine AldrichCPR](/deepweb/assets/sigmaaldrich/product/structures/187/001/4862c14e-bec7-4475-85a5-f178e48ff60f/640/4862c14e-bec7-4475-85a5-f178e48ff60f.png)