Alle Fotos(2)
Wichtige Dokumente
126020
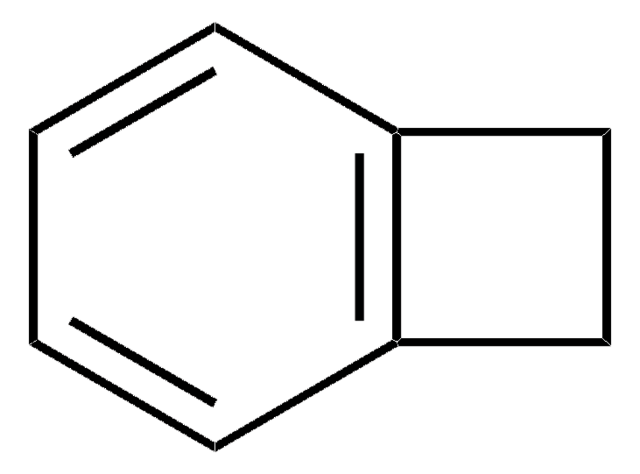
1,4-Dichlorphthalazin
95%
Anmeldenzur Ansicht organisationsspezifischer und vertraglich vereinbarter Preise
Alle Fotos(2)
About This Item
Empirische Formel (Hill-System):
C8H4Cl2N2
CAS-Nummer:
Molekulargewicht:
199.04
EG-Nummer:
MDL-Nummer:
UNSPSC-Code:
12352100
PubChem Substanz-ID:
NACRES:
NA.22
Empfohlene Produkte
Qualitätsniveau
Assay
95%
mp (Schmelzpunkt)
160-162 °C (lit.)
Funktionelle Gruppe
chloro
Lagertemp.
−20°C
SMILES String
Clc1nnc(Cl)c2ccccc12
InChI
1S/C8H4Cl2N2/c9-7-5-3-1-2-4-6(5)8(10)12-11-7/h1-4H
InChIKey
ODCNAEMHGMYADO-UHFFFAOYSA-N
Verwandte Kategorien
Anwendung
1,4-Dichlorophthalazine was used as starting reagent in the synthesis of series of 4-aryl-1-(4-methylpiperazin-1-yl)phthalazines. It was used as coupling reagent in the synthesis of novel soluble polymer-bound ligand. It was often used as building block in medicinal chemistry synthesis.
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Persönliche Schutzausrüstung
Eyeshields, Gloves, type N95 (US)
Hier finden Sie alle aktuellen Versionen:
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
K De Wael et al.
Science & justice : journal of the Forensic Science Society, 55(6), 422-430 (2015-12-15)
Reactively-dyed black, navy blue and medium red cotton samples showing metamerism under fluorescent tube illumination were examined. Optical microscopy (bright field, polarization and fluorescence microscopy) was used, followed by microspectrometry in the visible range (MSP Vis), to differentiate the samples
Synthesis of 4-aryl-1-(4-methylpiperazin-1-yl) phthalazines by Suzuki-type cross-coupling reaction.
Guery S, et al.
Synthesis, 2001(05), 699-701 (2001)
Matthew A J Duncton et al.
Bioorganic & medicinal chemistry letters, 16(6), 1579-1581 (2006-01-03)
A novel class of 1-(isoquinolin-5-yl)-4-arylamino-phthalazines is described as inhibitors of vascular endothelial growth factor receptor II (VEGFR-2). Many compounds display VEGFR-2 inhibitory activity with an IC(50) as low as 0.017 microM in an HTRF enzymatic assay. The compounds also inhibit
A simple and effective soluble polymer-bound ligand for the asymmetric dihydroxylation of olefins: DHQD-PHAL-OPEG-OMe.
Kuang YQ, et al.
Tetrahedron Letters, 42(34), 5925-5927 (2001)
William H Bunnelle et al.
Journal of medicinal chemistry, 50(15), 3627-3644 (2007-06-26)
A series of exceptionally potent agonists at neuronal nicotinic acetylcholine receptors (nAChRs) has been investigated. Several N-(3-pyridinyl) derivatives of bridged bicyclic diamines exhibit double-digit-picomolar binding affinities for the alpha 4 beta 2 subtype, placing them with epibatidine among the most
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.