ROAMYGL
Roche
Amyloglucosidase
from Aspergillus niger
Synonym(s):
Glucoamylase, disaccharidase-type-α-D-glucosidase
About This Item
Recommended Products
biological source
Aspergillus niger
Quality Level
form
suspension
specific activity
~14 units/mg protein (At 25 °C with glycogen as the substrate; standardized with BSA.)
mol wt
Mr 97 kDa
packaging
pkg of 10 mL (10102857001 [100 mg])
manufacturer/tradename
Roche
parameter
55 °C optimum reaction temp.
optimum pH
4.6-4.8
storage temp.
2-8°C
Related Categories
General description
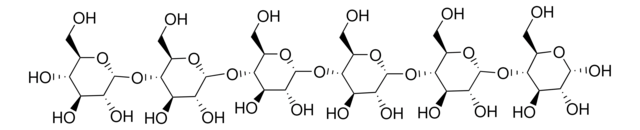
Specificity
Heat inactivation: Heat inactivation is recommended at 80 °C for 45 minutes, followed by rapidly cooling down.
Application
Biochem/physiol Actions
Unit Definition
Physical form
Other Notes
Storage Class
12 - Non Combustible Liquids
wgk_germany
WGK 1
flash_point_f
does not flash
flash_point_c
does not flash
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service