All Photos(1)
About This Item
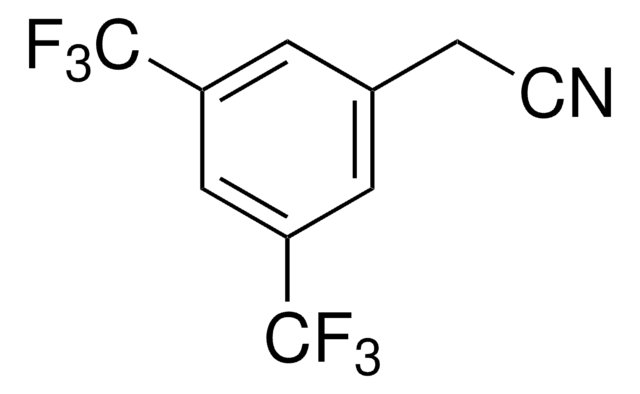
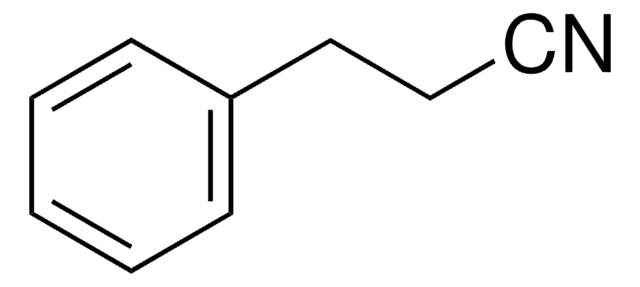
Linear Formula:
CF3C6H4CH2CN
CAS Number:
Molecular Weight:
185.15
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
form
solid
bp
131-132 °C/20 mmHg (lit.)
mp
47-49 °C (lit.)
SMILES string
FC(F)(F)c1ccc(CC#N)cc1
InChI
1S/C9H6F3N/c10-9(11,12)8-3-1-7(2-4-8)5-6-13/h1-4H,5H2
InChI key
QNKOCFJZJWOXDE-UHFFFAOYSA-N
Related Categories
Application
4-(Trifluoromethyl)phenylacetonitrile was used in the preparation of:
- 1,4-bis[2-cyano-2-(4-(trifluoromethyl)phenyl)vinyl]benzene
- cyano-substituted distyrylbenzene derivative, novel n-type organic semiconductor
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
235.4 °F - closed cup
Flash Point(C)
113 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Immacolata Serra et al.
Marine biotechnology (New York, N.Y.), 21(2), 229-239 (2019-01-27)
A screening among marine yeasts was carried out for nitrile hydrolyzing activity. Meyerozyma guilliermondii LM2 (UBOCC-A-214008) was able to efficiently grow on benzonitrile and cyclohexanecarbonitrile (CECN) as sole nitrogen sources. A two-step one-pot method for obtaining cells of M. guilliermondii
Synthesis and FET characteristics of phenylene-vinylene and anthracene-vinylene compounds containing cyano groups.
Shoji K, et al.
Journal of Materials Chemistry, 20(31), 6472-6478 (2010)
A Steady Operation of n-Type Organic Thin-Film Transistors with Cyano-Substituted Distyrylbenzene Derivative.
Nagamatsu S, et al.
Applied Physics Express, 2(10), 101502-101502 (2009)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service