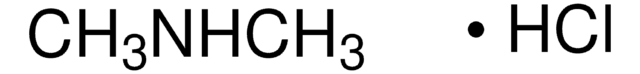PHR1084
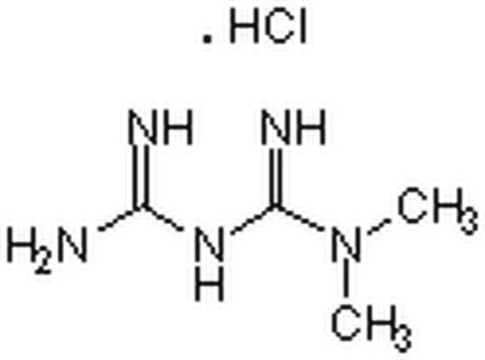
Metformin hydrochloride
Pharmaceutical Secondary Standard; Certified Reference Material
Synonym(s):
1,1-Dimethylbiguanide hydrochloride, Metformin
About This Item
Recommended Products
grade
certified reference material
pharmaceutical secondary standard
Quality Level
Agency
traceable to Ph. Eur. M0605000
traceable to USP 1396309
API family
metformin
CofA
current certificate can be downloaded
packaging
pkg of 500 mg
technique(s)
HPLC: suitable
gas chromatography (GC): suitable
mp
223-226 °C (lit.)
application(s)
pharmaceutical (small molecule)
format
neat
storage temp.
2-8°C
SMILES string
Cl[H].CN(C)C(=N)NC(N)=N
InChI
1S/C4H11N5.ClH/c1-9(2)4(7)8-3(5)6;/h1-2H3,(H5,5,6,7,8);1H
InChI key
OETHQSJEHLVLGH-UHFFFAOYSA-N
Gene Information
Looking for similar products? Visit Product Comparison Guide
General description
Application
Biochem/physiol Actions
Analysis Note
Other Notes
Footnote
Recommended products
analytical column
recommended
solvent
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Sorry, we don't have COAs for this product available online at this time.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Protocols
Separation of Melamine; 1,1-Dimethylbiguanide hydrochloride
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service