PHR1490
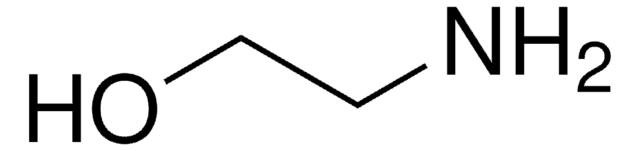
Monoethanolamine
Pharmaceutical Secondary Standard; Certified Reference Material
Sinónimos:
Ethanolamine, 2-Aminoethanol, 2-Aminoethyl alcohol, ETA, MEA, MEA 90, MEA-LCI, Monoethanolamine
About This Item
Productos recomendados
grade
certified reference material
pharmaceutical secondary standard
Quality Level
agency
traceable to Ph. Eur. Y0001184
traceable to USP 1445925
vapor density
2.1 (vs air)
vapor pressure
0.2 mmHg ( 20 °C)
description
Pharmaceutical Secondary Standard; Certified Reference Material
API family
ethanolamine
CofA
current certificate can be downloaded
autoignition temp.
1436 °F
expl. lim.
17 %
packaging
pkg of 1 mL
technique(s)
HPLC: suitable
gas chromatography (GC): suitable
refractive index
n20/D 1.454 (lit.)
bp
170 °C (lit.)
69-70 °C/10 mmHg
mp
10-11 °C (lit.)
density
1.012 g/mL at 25 °C (lit.)
application(s)
pharmaceutical (small molecule)
format
neat
storage temp.
2-30°C
SMILES string
NCCO
InChI
1S/C2H7NO/c3-1-2-4/h4H,1-3H2
InChI key
HZAXFHJVJLSVMW-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
General description
Application
Analysis Note
Other Notes
Footnote
Recommended products
related product
signalword
Danger
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Aquatic Chronic 3 - Eye Dam. 1 - Skin Corr. 1B - STOT SE 3
target_organs
Respiratory system
Storage Class
8A - Combustible corrosive hazardous materials
wgk_germany
WGK 2
flash_point_f
195.8 °F - Pensky-Martens closed cup
flash_point_c
91 °C - Pensky-Martens closed cup
Elija entre una de las versiones más recientes:
Certificados de análisis (COA)
It looks like we've run into a problem, but you can still download Certificates of Analysis from our Documentos section.
Si necesita más asistencia, póngase en contacto con Atención al cliente
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico