Black Hole Quencher™
Two commonly used quenchers, TAMRA™ and DABYCL, limit the ultimate sensitivity and flexibility of qPCR. TAMRA is not a dark quencher and therefore contributes to an overall increase in background because of its own native fluorescence. DABCYL, though a dark quencher, has an inadequate absorption footprint that overlaps very poorly with reporter dyes emitting above 480 nm (Figure 2).
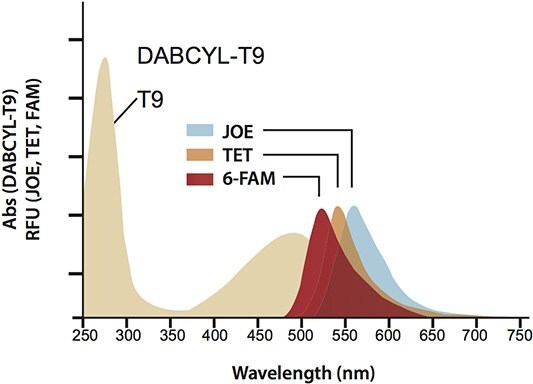
Figure 2.Absorption spectra of DABCYL-T9 with the emission spectra of the commonly-used reporters 6-FAM™, TET™, and JOE™. DABCYL was normalized at the poly-T absorbance (260 nm). RFU = relative fluorescence unit.
Though DABYCL works well in Molecular Beacons (structured probes with paired reporter and quencher that utilize static quenching), its absorption maximum of 474 nm places it below the maxima of the reporters shown in Figure 2, therefore it is a poor choice for Dual-Labeled Probes (linear probes with separated reporter and quencher that utilize FRET quenching).
Black Hole Quencher™ 1 (BHQ™-1) (with an absorption maximum of 534 nm) is absorbed at higher wavelengths than DABCYL and is directly superimposable with the emission maxima of FAM, TET and JOE. This provides a significant increase in both static and FRET quenching efficiencies, which makes it an excellent quencher for Molecular Beacons and Dual-Labeled Probes. As shown in Figure 3, BHQ™-1, BHQ™-2, and BHQ™-3 cover the spectrum from 480 nm into the near IR making it possible to utilize reporters that emit anywhere in this range.
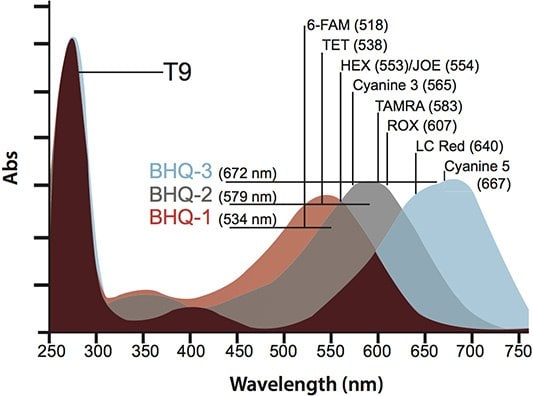
Figure 3.Absorption spectra of the three BHQ™ variants (conjugated to T-9 and normalized to the poly-T absorbance of 260 nm) with the emission maxima of the commonly-used reporters 6-FAM, TET, HEX™, Cyanine 3, TAMRA, ROX™, LC Red 640, and Cyanine 5.
In addition to better spectral overlap, BHQ™ probes have much higher signal-to-noise ratios when compared to the corresponding DABCYL or TAMRA probes (Figure 4).
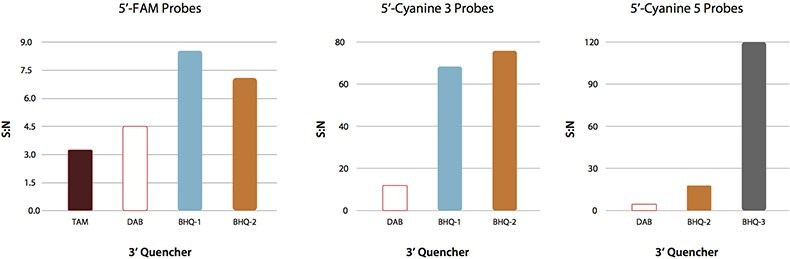
Figure 4.Signal-to-noise (S:N) ratios were calculated by dividing the fluorescence signal of a 25mer in the presence of a five-fold excess of an exact complementary target sequence by the fluorescence intensity of the probe alone.
Finally, as shown in Figure 5, the BHQ™ variants readily permit single-tube multiplexing due to the increased variety of reporters that can be effectively quenched with little or no cross-talk. This simplifies the design, implementation, and interpretation of multiplexed assays.
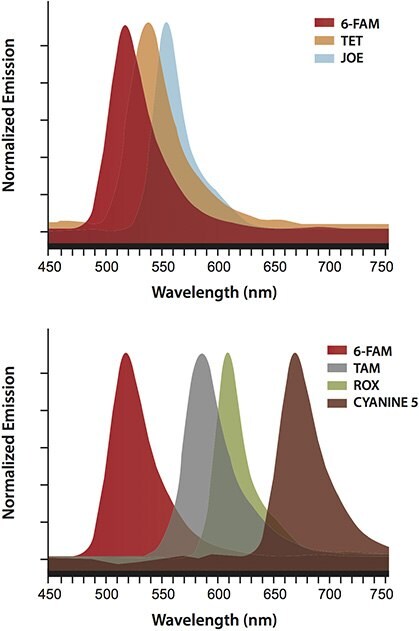
Figure 5.The unique characteristics of the BHQ™ variants permit flexibility in the choice of spectrally well-resolved reporters, which enable single-tube multiplexing with little or no cross-talk.
In summary, Black Hole Quencher™:
- Has no native fluorescence (emits heat instead of light), resulting in lower background fluorescence
- Has increased signal-to-noise ratio, providing higher sensitivity
- Enables a wider choice of reporters for multiplexing
To continue reading please sign in or create an account.
Don't Have An Account?