C-O Cross-Coupling of Activated Aryl and Heteroaryl Halides with Aliphatic Alcohols
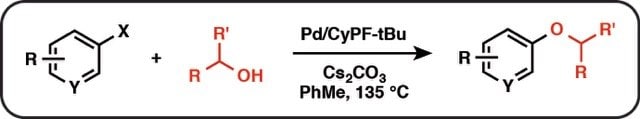
Figure 1.
Abstract
We report an unprecedented BrettPhos ligand supported Pd-catalyzed C-O bond-forming reaction of activated aryl halides with primary fluoroalkyl alcohols. We demonstrate that the Phosphine ligand (BrettPhos) possesses the property of altering the mechanistic pathway of reductive elimination from nucleophile to nucleophile. The Pd/BrettPhos catalyst system facilitates the reductive elimination of the oxygen nucleophile through an electronic pathway.
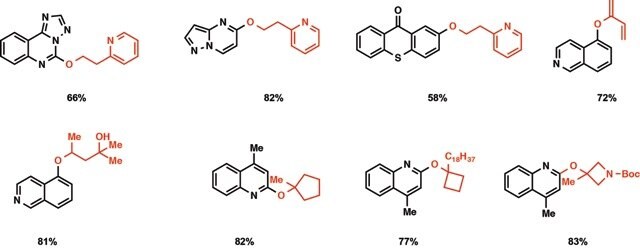
Figure 2.
Procedure
An oven dried 5.0 mL two-neck round-bottomed flask was equipped with a magnetic stir bar, a rubber septum, condenser, and an argon balloon on the top of the condenser with the aid of an adaptor. The flask was charged with Cs2CO3 and dried with hot air gun under vacuum. The R.B. flask was allowed to cool under an argon atmosphere. [Pd2(dba)3] and BrettPhos ligand were added successively. The flask was then flushed with argon repeatedly three times. To this, anhydrous toluene (1.0 mL) was added by sy- ringe and the mixture was stirred for five minutes at room temperature. A solution of aryl halides and fluoroalcohols in toluene (1.0 mL) was added to the flask by syringe. Additional toluene (1.0 mL, for rinsing) was added to the reaction flask and the flask was placed into a pre-heated oil bath at 85 °C. The reaction mixture was stirred vigorously until completion as indicated by TLC analysis. The reaction mixture was allowed to cool to room temperature and passed through a short silica (230 400 mesh size) column eluted with chloroform or directly with hexane/ethyl acetate solvent mixture under N2 pressure. The solvent removal under reduced pressure afforded the desired compounds as a yellow oily liquid or yellow/colorless solid.
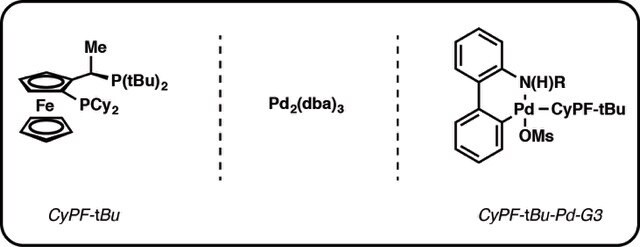
Figure 3.
References
To continue reading please sign in or create an account.
Don't Have An Account?