HumanKine® Thermostable bFGF, an engineered recombinant protein with enhanced biological functionality on human iPS and neural progenitor cells
Nick Asbrock, Christine Chen, Vi Chu
MilliporeSigma, Temecula, CA, USA
Introduction
Fibroblast growth factors (FGFs) are secreted glycoproteins that regulate several fundamental developmental pathways and help regulate mesoderm and ectoderm patterning in the early embryonic development. In adults, FGFs participate in several physiological and pathological processes including cell proliferation, cell survival, differentiation, wound healing, and regulation of angiogenesis. The FGF signaling process involves their binding to FGF receptors (FGFR) on the cell surface. Twenty-three different FGFs have been identified that exert their effects through four major types of tyrosine kinase-linked FGFRs that are designated as FGFR1, FGFR2, FGFR3, and FGFR4. Following ligand binding, these receptors undergo dimerization, which results in the activation of their tyrosine kinase domains and intermolecular transphosphorylation.
Basic FGF, also known as bFGF or FGF-2, belongs to a group of structurally homologous FGFs that are characterized by heparin binding and mitogenic activity on a variety of cells of mesodermal and neuroectodermal origin. The use of bFGF in stem cell biology is widespread and is a critical growth factor that enables the expansion of many stem cells in their pluripotent or multipotent states. Several recent studies suggest an important role for FGF/extracellular signal-regulated kinase (Erk) signaling in promoting the transition from a naïve to a primed differentiation state and in preventing primed cells from reverting back to a naïve state. FGF signaling, in effect, stabilizes the primed cell state of human embryonic stem cells. Due to its rapid degradation at 37 °C, many stem cell researchers add high levels of bFGF to their media and require cumbersome frequent media exchanges.
The HumanKine® Thermostable bFGF is an engineered recombinant version of human bFGF. It is generated by selectively altering the amino acid sequence to enhance its thermostability compared to wild-type bFGF. This humanized recombinant protein is produced using a proprietary human cell expression system and is designated as a xeno-free product. We have shown that this unique protein has enhanced stability at 37 °C while possessing increased biological functionality on both human iPS cells and human neural stem cell populations. The use of this product will reduce the variation commonly observed within many stem cell cultures and will reduce the time and cost needed to culture these demanding cells.
Methods
Determining stability of bFGF cytokines at elevated temperatures
The stability of normal and HumanKine® Thermostable bFGF recombinant proteins was analyzed at 37 °C over a period of 7 days. Normal bFGF or HumanKine® Thermostable bFGF was diluted in 20% KOSR medium to a final concentration of 150 ng/mL. Samples were allowed to remain at 37 °C over a period of one week with stopping at different time points during this period. The level of remaining bFGF was quantified at each time point using a quantitative bFGF ELISA kit.
Culture of human iPS cells
Human iPS cells were generated from neonatal human foreskin fibroblasts using STEMCCA™ lentivirus reprogramming kit followed by excision of transgenes using a cell-permeable Cre-Recombinase. Transgene-free iPS cells were maintained in KOSR medium with standard bFGF (10 ng/mL) on primary mouse embryonic fibroblasts for 20 passages using a manual passaging technique until used for experiments. At passage 21, Human iPS cells were manually passed into either KOSR medium with no FGF, KOSR + Normal bFGF (10 ng/mL) or KOSR medium + Thermostable bFGF (10 ng/mL) conditions in duplicate at approximately 50 colonies/well with feeder cells present. Cells were fed every third day for one passage under above conditions and were fixed and exposed to anti-Oct-4 and anti-TRA-1-60 for immunostaining. The total number of differentiated colonies were manually counted on day 7.
Expansion of ENStem™-A human neural progenitors
ENStem™-A cells at passage 20 were thawed onto Matrigel matrix-coated plates (1:50) in ENStem™-A neural expansion medium with standard bFGF (20 ng/mL) or HumanKine® Thermostable bFGF (20 ng/mL). Cells were passaged using Accutase™ reagent and, when 80% confluency was reached for three passages, they were immunostained with neural stem cell markers anti-nestin and anti-Sox-2.
To assess proliferation, 5x104 cells were seeded onto Matrigel matrix-coated (1:50) 6-well plates in duplicate in either ENStem™-A neural expansion medium + standard bFGF (20 ng/mL) or ENStem™-A neural expansion medium + Thermostable bFGF (20 ng/mL). Cells were allowed to grow for 6 days without any media exchanges. Cells were detached and triplicate samples were mixed with Trypan blue and counted using a hemocytometer at days 2, 4, and 6.
Results
Thermostability Experiments
We studied the thermal stability of normal bFGF and modified, thermostable bFGF at 37 °C over a period of seven days (Figure 2). The reduction in normal bFGF levels was more pronounced (nearly 95% reduction on day 1) compared to the thermostable bFGF (only ~60% reduction on day 1). Both bFGFs showed gradual decrease over a period of 7 days; however, at a slower rate of decline with the thermostable bFGF.

Figure 2. Enhanced stability of thermostable bFGF at 37 °C vs. normal bFGF.
Human iPS cell experiments
When we used a limited feeding schedule to culture human iPS cells and used thermostable bFGF, we observed elevated levels of the pluripotency markers Oct-4 and TRA-1-60 after 7 days and significant reduction in spontaneous differentiation of human iPS cells compared to when we used normal bFGF in the same feeding schedule (Figures 3 and 4). In fact, we found no significant reduction in the number of spontaneously differentiated colonies when using normal bFGF compared to KOSR medium alone (Figure 4).
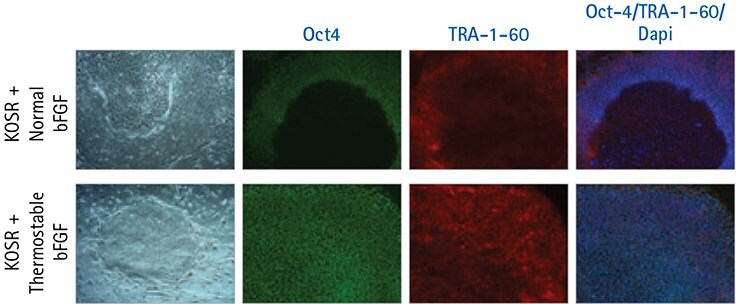
Figure 3. Thermostable bFGF supports pluripotent growth of human iPS cells using an every-third-day feeding regiment.
Human iPS cells grown in HumanKine® Thermostable bFGF following a limited feeding regimen express elevated levels of the pluripotency markers Oct-4 (Green) and TRA-1-60 (Red) after 7 days. Human IPS cells grown with KOSR medium with normal bFGF following a limited feeding regimen spontaneously differentiate within the center of the colonies and do not express the same uniform levels of Oct-4 and TRA-1-60 expression.

Figure 4. Human iPS cells show lower levels of spontaneous differentiation using an every-third-day feeding regimen when used in combination with HumanKine® Thermostable bFGF.
ENStem™-A human neural stem cell experiments
To determine if thermostable bFGF could also facilitate the culture of neural progenitor cells, we first cultured them with thermostable bFGF using a normal feeding schedule (Figure 5). Immunocytochemistry and brightfield microscopy showed that the cells, after three passages, displayed proper morphology and expression of neural stem cell markers Sox-2 and nestin.

Figure 5. ENStem™-A neural progenitor cells grown for 3 passages in HumanKine® Thermostable bFGF following a normal feeding regimen display proper morphology and express the neural stem cell markers Sox-2 and Nestin. Nestin-stained cells were counterstained with DAPI (blue) nuclear stain.
Next, we measured proliferation of neural progenitor cells while keeping them on a minimal feeding schedule, in which cells were incubated for six days without any intervening medium changes. After six days, there were slightly more cells per well in the cultures containing thermostable bFGF as compared to normal bFGF (Figure 6).

Figure 6. Higher cell proliferation rates of ENStem™-A human neural progenitor cells using a limited feeding regimen when using thermostable bFGF compared to normal bFGF. (A) Time course showing cell counts on days 2, 4 and 6 of incubation. (B) Comparing cell number per well after six days of incubation without medium exchange. Experiments were performed using three replicates of each time point and culture condition.
Discussion
Fibroblast growth factor is known to play a key role in maintaining embryonic stem cells and iPS cells in a proliferative, undifferentiated state. Basic FGF is generally added to the culture medium to sustain ERK phosphorylation and Nanog expression1. bFGF levels required to support pluripotency over a period of time are about 10-fold higher than those required for other cell types2. Chen et al have shown that acidic FGF is highly unstable at 37 °C and fails to maintain ERK phosphorylation and pluripotency in stem cells1. A similar phenomenon was suggested for bFGF. However, their studies have indicated that, at 37 °C, bFGF undergoes aggregation, thereby reducing its biological availability for maintaining pluripotency.
Consistent with these indications, our data suggest that HumanKine® Thermostable bFGF can be used with human ESC, iPS and neural stem cells to maintain them in a pluripotent and multipotent state as a more effective alternative to normal bFGF. Furthermore, because of its increased stability, thermostable bFGF permits a less stringent feeding schedule, increasing the experimental flexibility of these cell models.
To continue reading please sign in or create an account.
Don't Have An Account?