SAMPHV001
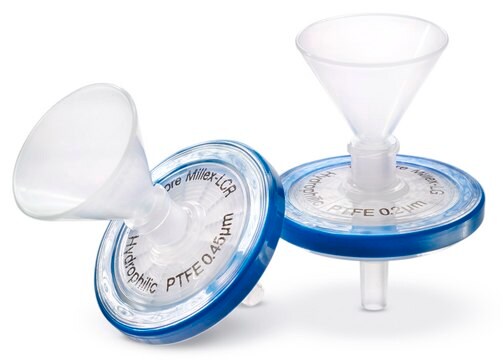
Millex Samplicity® filter
PVDF membrane, pore size 0.45 μm, diam. 25 mm, non-sterile, hydrophilic
Synonym(s):
0.45 μm PVDF syringe filter, Durapore® PVDF syringe filter, Millex-HV syringe filter, disposable syringe filter, syringe filter
About This Item
high-density polyethylene
Recommended Products
material
PVDF membrane
high-density polyethylene
Quality Level
sterility
non-sterile
product line
Millex®
feature
hydrophilic
manufacturer/tradename
Samplicity®
technique(s)
HPLC: suitable
diam.
25 mm
volume
1.7 mL
pore size
0.45 μm pore size
fitting
inlet funnel (Easy loading)
outlet tip (for smooth transfer to HPLC vials)
shipped in
ambient
General description
• Samplicity G2 Filtration System enables vacuum driven filtration of up to 8 samples at once, directly into HPLC vials.
• Filtration for HPLC sample prep without syringes
• Millex Samplicity Filters provide integrated funnels and filters for easiest sample loading
• Available in 0.20 µm and 0.45 µm pore sizes with hydrophilic PTFE and Durapore PVDF membranes
Available in 2 Pack Sizes:
SKU ending with 01 (-------01): 96 per pack
SKU ending with 04 (-------04): 384 devices (4x 96 per pack)
Applications:
HPLC; Filtration of viscous or particulate laden samples for HPLC
Application
Legal Information
Not finding the right product?
Try our Product Selector Tool.
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Related Content
This is an Application Note for multisample filtration of small volumes using the Samplicity® system
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service