Sildenafil Citrate Method (HPLC) following USP Monograph on Ascentis® Express C18 with UV detection
Sildenafil, sold under the brand name Viagra, among others, is a medication used to treat erectile dysfunction and pulmonary arterial hypertension. In 1994, Pfizer filed a patent covering the use of sildenafil to treat erectile dysfunction. This patent will expire in 2019.
Ascentis® Express C18 columns can be used for the HPLC methods within the sildenafil citrate USP Monograph, here illustrated with appropriate analytical data to verify that system suitability criteria are met per USP41-NF36 guidelines.
Same experimental conditions are used for both assay and organic impurities (related substances) but in the latter method the chromatograph should run for three (3) times the retention time of sildenafil. The suitability requirements for the assay method are defined reproducibility (not more than 0.85% for six replicates) and peak tailing factor (not more than 1.5) whereas for organic impurities the relative retention times for sildenafil, sildenafil N-oxide, and sildenafil related compound A are about 1.0, 1.2, and 1.7, respectively. The resolution should be not less than 2.5 between sildenafil N-oxide and sildenafil (using the system suitability solution), and the signal-to-noise ratio not less than 10 (using the sensitivity solution).
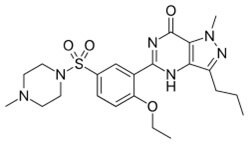
Sildenafil Citrate




To continue reading please sign in or create an account.
Don't Have An Account?