E24905
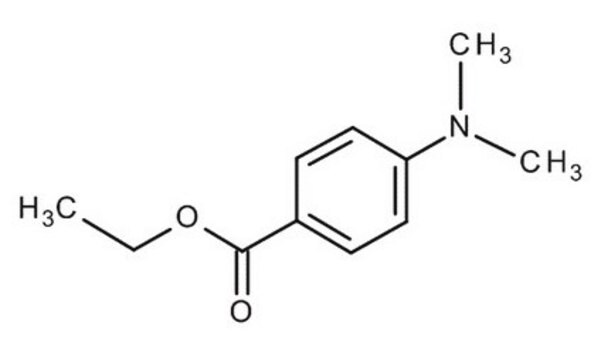
Ethyl 4-(dimethylamino)benzoate
≥99%
Synonym(s):
Parbenate
About This Item
Recommended Products
Quality Level
assay
≥99%
mp
63-66 °C (lit.)
SMILES string
CCOC(=O)c1ccc(cc1)N(C)C
InChI
1S/C11H15NO2/c1-4-14-11(13)9-5-7-10(8-6-9)12(2)3/h5-8H,4H2,1-3H3
InChI key
FZUGPQWGEGAKET-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
signalword
Danger
hcodes
Hazard Classifications
Aquatic Chronic 2 - Repr. 1B
Storage Class
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
With dentists placing nearly 100 million dental fillings into patients′ teeth annually in the U.S. alone, polymeric composite restoratives account for a very large share of the biomaterials market.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service