推薦產品
品質等級
化驗
≥98% (HPLC)
形狀
powder
起源
Takeda
SMILES 字串
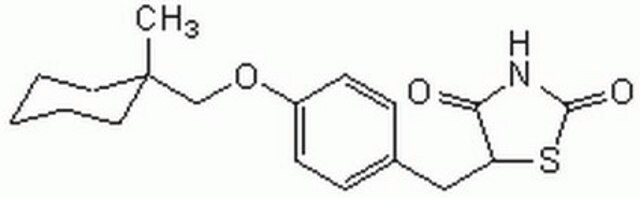
CC1(CCCCC1)COc2ccc(CC3SC(=O)NC3=O)cc2
InChI
1S/C18H23NO3S/c1-18(9-3-2-4-10-18)12-22-14-7-5-13(6-8-14)11-15-16(20)19-17(21)23-15/h5-8,15H,2-4,9-12H2,1H3,(H,19,20,21)
InChI 密鑰
YZFWTZACSRHJQD-UHFFFAOYSA-N
基因資訊
human ... PPARG(5468)
mouse ... Pparg(19016)
應用
Ciglitizone has been used as a proliferator-activated receptor γ (PPARγ) agonist:
It also may be used to study its effects on cell cycle and apoptosis in monocytic cells.
- to study its effects on cell proliferation in human melanocytes
- to study its effects on pigmentation and migration of human melanocytes
It also may be used to study its effects on cell cycle and apoptosis in monocytic cells.
生化/生理作用
Ciglitizone belongs to the class of thiazolidinediones and is a peroxisome proliferator-activated receptor γ (PPARγ) agonist. It exhibits anti-diabetic activity. Ciglitizone has the potential to treat tumor necrosis factor α (TNFα)-related apoptosis-inducing ligand (TRAIL)-refractory high-grade urothelial cancers.
特點和優勢
This compound is a featured product for Gene Regulation research. Click here to discover more featured Gene Regulation products. Learn more about bioactive small molecules for other areas of research at sigma.com/discover-bsm.
This compound was developed by Takeda. To browse the list of other pharma-developed compounds and Approved Drugs/Drug Candidates, click here.
儲存類別代碼
10 - Combustible liquids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
Eyeshields, Gloves, multi-purpose combination respirator cartridge (US)
分析證明 (COA)
輸入產品批次/批號來搜索 分析證明 (COA)。在產品’s標籤上找到批次和批號,寫有 ‘Lot’或‘Batch’.。
客戶也查看了
Eun Jong Han et al.
International journal of radiation oncology, biology, physics, 85(5), e239-e248 (2013-01-22)
To investigate possible radiosensitizing activities of the well-known peroxisome proliferator-activated receptor (PPAR)γ ligand ciglitazone and novel PPARγ ligands CAY10415 and CAY10506 in non-small cell lung cancer (NSCLC) cells. Radiosensitivity was assessed using a clonogenic cell survival assay. To investigate the
S Zulkafli Nor Effa et al.
Biomolecules, 8(4) (2018-11-08)
Immunomodulation, as a means of immunotherapy, has been studied in major research and clinical laboratories for many years. T-Regulatory (Treg) cell therapy is one of the modulators used in immunotherapy approaches. Similarly, nuclear receptor peroxisome proliferator activated receptor gamma (PPARγ)
The structure-activity relationship between peroxisome proliferator-activated receptor gamma agonism and the antihyperglycemic activity of thiazolidinediones.
T M Willson et al.
Journal of medicinal chemistry, 39(3), 665-668 (1996-02-02)
H Y Kang et al.
The British journal of dermatology, 150(3), 462-468 (2004-03-20)
Peroxisome proliferator-activated receptors (PPARs) belong to the superfamily of nuclear receptors that heterodimerize with the retinoic X receptor. Agonists of PPAR have been known to play an important role in cellular responses including proliferation and differentiation. The expression and function
[[omega-(Heterocyclylamino)alkoxy]benzyl]-2,4-thiazolidinediones as potent antihyperglycemic agents.
B C Cantello et al.
Journal of medicinal chemistry, 37(23), 3977-3985 (1994-11-11)
A series of [(ureidoethoxy)benzyl]-2,4-thiazolidinediones and [[(heterocyclylamino)alkoxy]-benzyl]-2,4-thiazolidinediones was synthesized from the corresponding aldehydes. Compounds from the urea series, exemplified by 16, showed antihyperglycemic potency comparable with known agents of the type such as pioglitazone and troglitazone (CS-045). The benzoxazole 49, a
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務