推薦產品
等級
certified reference material
pharmaceutical secondary standard
品質等級
agency
traceable to BP 999
traceable to Ph. Eur. I0150000
traceable to USP 1338801
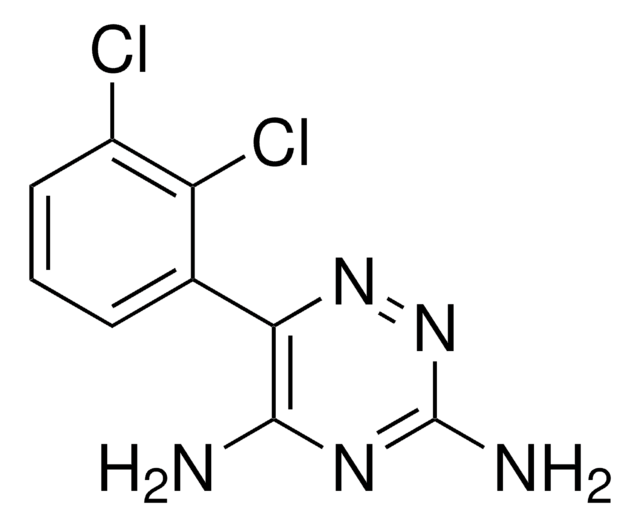
API 家族
indapamide
CofA
current certificate can be downloaded
包裝
pkg of 1 g
技術
HPLC: suitable
gas chromatography (GC): suitable
應用
pharmaceutical (small molecule)
形式
neat
儲存溫度
2-30°C
SMILES 字串
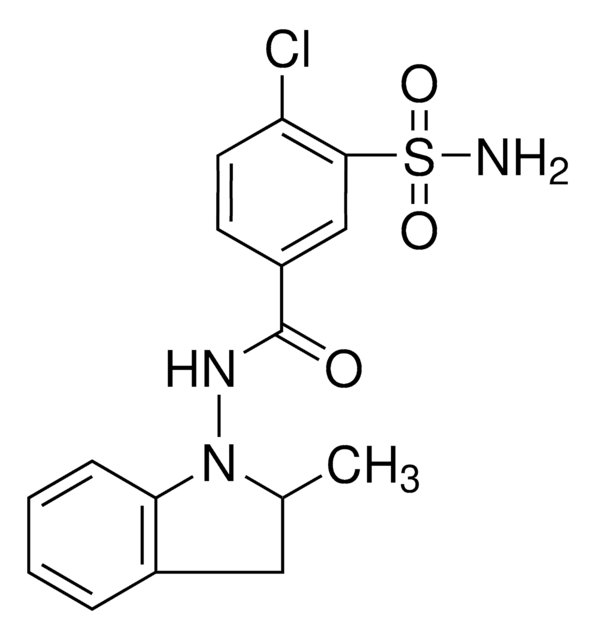
CC1Cc2ccccc2N1NC(=O)c3ccc(Cl)c(c3)S(N)(=O)=O
InChI
1S/C16H16ClN3O3S/c1-10-8-11-4-2-3-5-14(11)20(10)19-16(21)12-6-7-13(17)15(9-12)24(18,22)23/h2-7,9-10H,8H2,1H3,(H,19,21)(H2,18,22,23)
InChI 密鑰
NDDAHWYSQHTHNT-UHFFFAOYSA-N
基因資訊
human ... SLC12A3(6559)
尋找類似的產品? 前往 產品比較指南
一般說明
Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.
Indapamide is a diuretic that is categorized under the family of benzothiadiazines compounds. It is an oral drug that is usually prescribed as an antihypertensive agent for patients with mild to moderate hypertension.
Indapamide is a diuretic that is categorized under the family of benzothiadiazines compounds. It is an oral drug that is usually prescribed as an antihypertensive agent for patients with mild to moderate hypertension.
應用
Indapamide may be used as a pharmaceutical reference standard for the determination of the analyte in pharmaceutical formulations by various chromatography techniques and spectrophotometry.
These Secondary Standards are qualified as Certified Reference Materials. These are suitable for use in several analytical applications including but not limited to pharma release testing, pharma method development for qualitative and quantitative analyses, food and beverage quality control testing, and other calibration requirements.
分析報告
These secondary standards offer multi-traceability to the USP, EP and BP primary standards, where they are available.
其他說明
This Certified Reference Material (CRM) is produced and certified in accordance with ISO 17034 and ISO/IEC 17025. All information regarding the use of this CRM can be found on the certificate of analysis.
相關產品
產品號碼
描述
訂價
訊號詞
Warning
危險聲明
危險分類
Lact. - Repr. 2
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 2
閃點(°F)
Not applicable
閃點(°C)
Not applicable
客戶也查看了
Determination of certain drugs in binary mixtures formulations by second derivative ratio spectrophotometry and LC.
El-Gindy A, et al.
Il Farmaco (Societa Chimica Italiana : 1989), 59(9), 703-712 (2004)
Development of new spectrophotometric methods for the determination of Indapamide in bulk and Pharmaceutical formulations.
Pai JB, et al.
International Journal of ChemTech Research, 3(2), 755-760 (2011)
Quantitative determination of indapamide in pharmaceuticals and urine by high-performance liquid chromatography with amperometric detection.
Legorburu MJ, et al.
Journal of Chromatographic Science, 37(8), 283-287 (1999)
Comparison of spectrophotometric and an LC method for the determination perindopril and indapamide in pharmaceutical formulations.
Erk N
Journal of Pharmaceutical and Biomedical Analysis, 26(1), 43-52 (2001)
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務