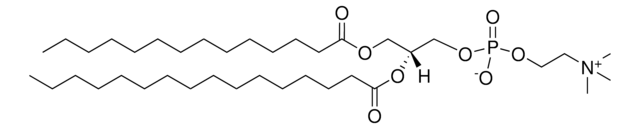
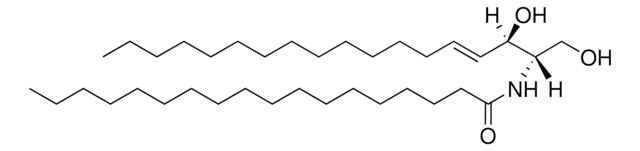
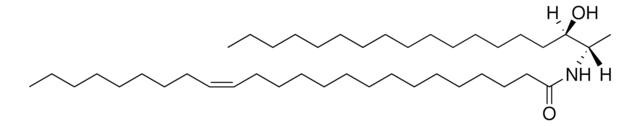
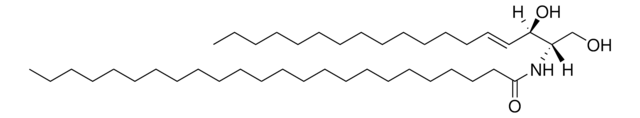
推薦產品
化驗
>99% (TLC)
形狀
powder
包裝
pkg of 1 × 1 mg (860462P-1mg)
pkg of 1 × 5 mg (860462P-5mg)
製造商/商標名
Avanti Research™ - A Croda Brand 860462P
脂質類型
sphingolipids
bioactive lipids
運輸包裝
dry ice
儲存溫度
−20°C
一般說明
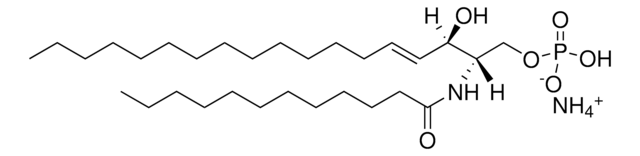
Commonly referred to as 1-deoxydihydroceramide (1-deoxyDHCer), this product is the N-acylated form of 1-deoxysphinganine, a potent inhibitor of sphingolipid metabolism. The biological activity of 1-deoxyDHCer is not clearly understood at this time.
N-C16-deoxysphinganine Commonly referred to as 1-deoxydihydroceramide (1-deoxyDHCer), is the N-acylated form of 1-deoxysphinganine, a potent inhibitor of sphingolipid metabolism. The N-acyl group can be 16, 20 and 24 carbon chain. N-acylsphinganines (dihydroceramides) are synthesized by the acylation of sphingoid bases in the presence of ceramide synthases (CerS).
生化/生理作用
N-acylsphinganines (dihydroceramides) levels are lower during ceramide synthase inhibition. They are intermediates of ceramide and dihydrosphingolipids synthesis. Treatment of MCF7 cancer cells with fenretinide alters sphingolipid metabolism.
包裝
5 mL Amber Glass Screw Cap Vial (860462P-1mg)
5 mL Amber Glass Screw Cap Vial (860462P-5mg)
法律資訊
Avanti Research is a trademark of Avanti Polar Lipids, LLC
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
Sphingolipidomics: a valuable tool for understanding the roles of sphingolipids in biology and disease
Merrill AH, et al.
Journal of Lipid Research, 50, S97-S102 (2009)
Ceramide synthase inhibition by fumonisin B1 causes accumulation of 1-deoxysphinganine a novel category of bioactive 1-deoxysphingoid bases and 1-deoxydihydroceramides biosynthesized by mammalian cell lines and animals
Zitomer NC, et al.
The Journal of Biological Chemistry, 284(8), 4786-4795 (2009)
Thematic Review Series: Sphingolipids. Biodiversity of sphingoid bases (?sphingosines?) and related amino alcohols
Pruett ST, et al.
Journal of Lipid Research, 49(8), 1621-1639 (2008)
Noemi Jiménez-Rojo et al.
Biophysical journal, 107(12), 2850-2859 (2014-12-18)
Ceramides and dihydroceramides are N-acyl derivatives of sphingosine and sphinganine, respectively, which are the major sphingoid-base backbones of mammals. Recent studies have found that mammals, like certain other organisms, also produce 1-deoxy-(dihydro)ceramides (1-deoxyDHCers) that contain sphingoid bases lacking the 1-hydroxyl-
Sarah T Pruett et al.
Journal of lipid research, 49(8), 1621-1639 (2008-05-24)
"Sphingosin" was first described by J. L. W. Thudichum in 1884 and structurally characterized as 2S,3R,4E-2-aminooctadec-4-ene-1,3-diol in 1947 by Herb Carter, who also proposed the designation of "lipides derived from sphingosine as sphingolipides." This category of amino alcohols is now
我們的科學家團隊在所有研究領域都有豐富的經驗,包括生命科學、材料科學、化學合成、色譜、分析等.
聯絡技術服務